Unit 5 - Notes
Unit 5: Corrosion
1. Definition and Scope of Corrosion
Definition:
Corrosion is defined as the gradual destruction or deterioration of a metal or alloy by the chemical or electrochemical reaction with its environment. It is essentially "metallurgy in reverse," where refined metals return to their native, stable ore states (oxides, sulfides, carbonates, etc.).
Scope and Significance:
- Economic Impact: Causes massive losses in direct replacement costs and maintenance.
- Safety: Leads to structural failures in bridges, aircraft, and pipelines.
- Contamination: Product contamination in food and pharmaceutical industries due to corroded equipment.
- Efficiency: Reduces the efficiency of heat exchangers and boilers due to scale formation.
2. Direct Chemical Corrosion (Dry Corrosion)
This type of corrosion occurs in the absence of moisture, usually involving a direct chemical attack by gases (Oxygen, Halogens, , ) on the metal surface at ambient or elevated temperatures.
Mechanism of Oxidation Corrosion
- Attack: Oxygen attacks the metal surface directly.
- Oxidation: The metal loses electrons:
- Reduction: Oxygen gains electrons:
- Scale Formation: Metal oxide forms on the surface ().
Nature of Oxide Films (Pilling-Bedworth Rule):
The protective nature of the film depends on the volume of the oxide formed () relative to the volume of metal consumed ().
- Stable/Protective: If (e.g., Al, Sn, Pb, Cu), the film is non-porous and prevents further corrosion.
- Unstable/Non-protective: If (e.g., Li, Na, K), the film is porous, allowing oxygen to penetrate and corrosion continues.
- Volatile: If the oxide evaporates (e.g., Molybdenum oxide), fresh metal is continuously exposed.
3. Electrochemical Corrosion (Wet Corrosion)
This occurs when a conducting liquid is in contact with metal or when two dissimilar metals are immersed or dipped partially in a solution. It involves the formation of anodic and cathodic areas.
General Mechanism:
- Anode: Oxidation occurs (loss of electrons). Metal dissolves.
- Cathode: Reduction occurs (gain of electrons). No corrosion of the cathode metal itself.
- Electron Flow: From Anode to Cathode through the metal.
- Ion Flow: Through the electrolyte.
Mechanism 1: Evolution of Hydrogen
- Occurs in acidic environments.
- Anode:
- Cathode: Hydrogen ions () from the acid gain electrons.
- Result: Displacement of hydrogen gas from the acidic solution.
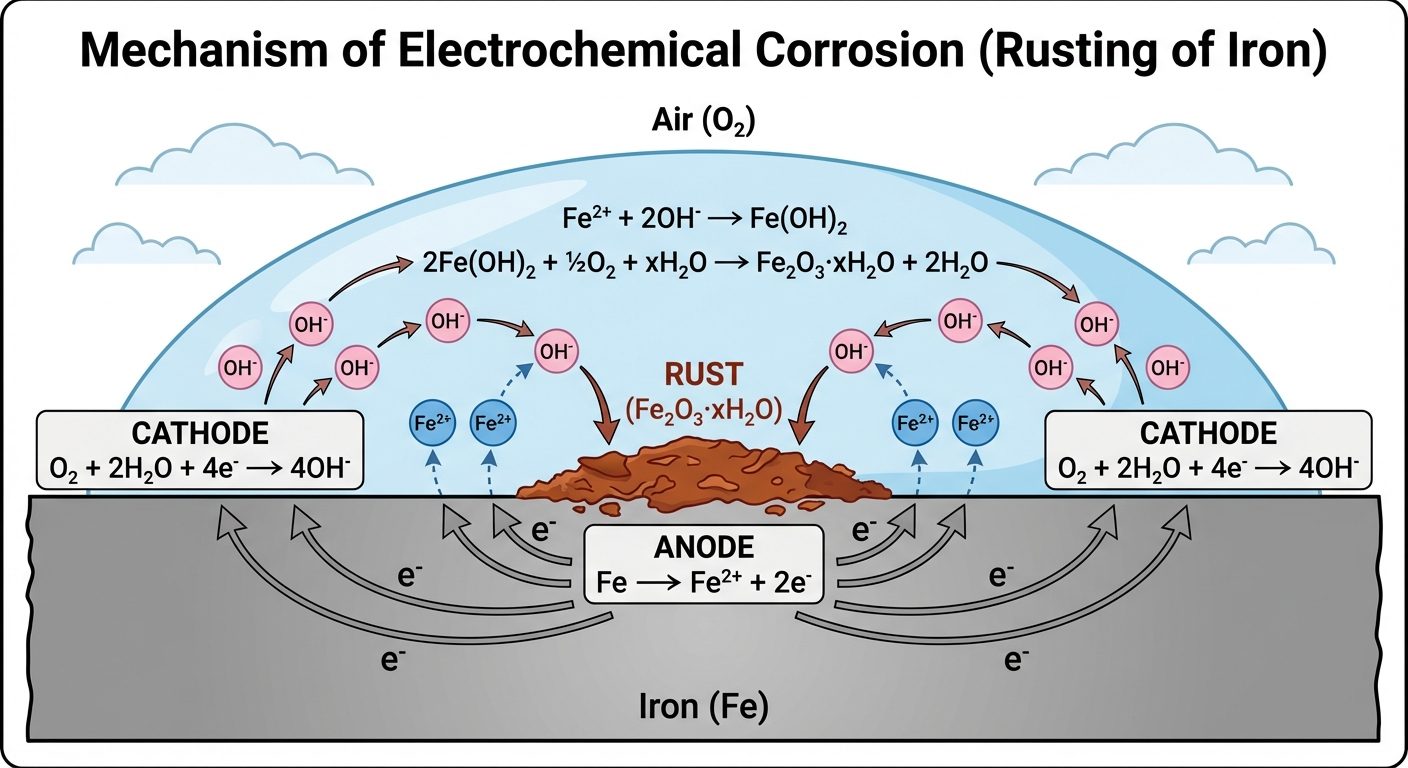
Mechanism 2: Absorption of Oxygen
- Occurs in neutral or alkaline environments (e.g., rusting of iron in water).
- Anode: (Corrosion occurs here).
- Cathode: Dissolved oxygen takes up electrons.
- Product: The and ions diffuse and combine to form Ferrous Hydroxide, which further oxidizes to Hydrated Ferric Oxide (Yellow/Red Rust).
- (Rust).
4. Types of Electrochemical Corrosion
A. Galvanic Corrosion (Bimetallic Corrosion)
Occurs when two dissimilar metals are electrically connected and exposed to an electrolyte.
- Principle: The metal higher in the Electrochemical Series (more active/negative reduction potential) acts as the Anode and corrodes. The nobler metal acts as the Cathode and is protected.
- Example: Zinc coupled with Copper. Zinc (Anode) corrodes; Copper (Cathode) remains safe.
- Control: Insulate the metals from each other or use a "sacrificial anode."
B. Concentration Cell Corrosion
Occurs even in a single metal if different parts of it are exposed to different concentrations of the electrolyte.
- Rule: The part of the metal in contact with the lower concentration of electrolyte becomes the Anode (corrodes). The part in higher concentration becomes the Cathode.
C. Differential Aeration Corrosion
This is a specific type of concentration cell corrosion based on oxygen exposure.
- Principle: "Poorly oxygenated parts become Anodic; highly oxygenated parts become Cathodic."
- Mechanism:
- Cathode (High ):
- Anode (Low ):
- Result: Corrosion occurs in crevices, under dirt piles, or partially immersed areas where oxygen supply is restricted.
5. Specific Forms of Differential Aeration Corrosion
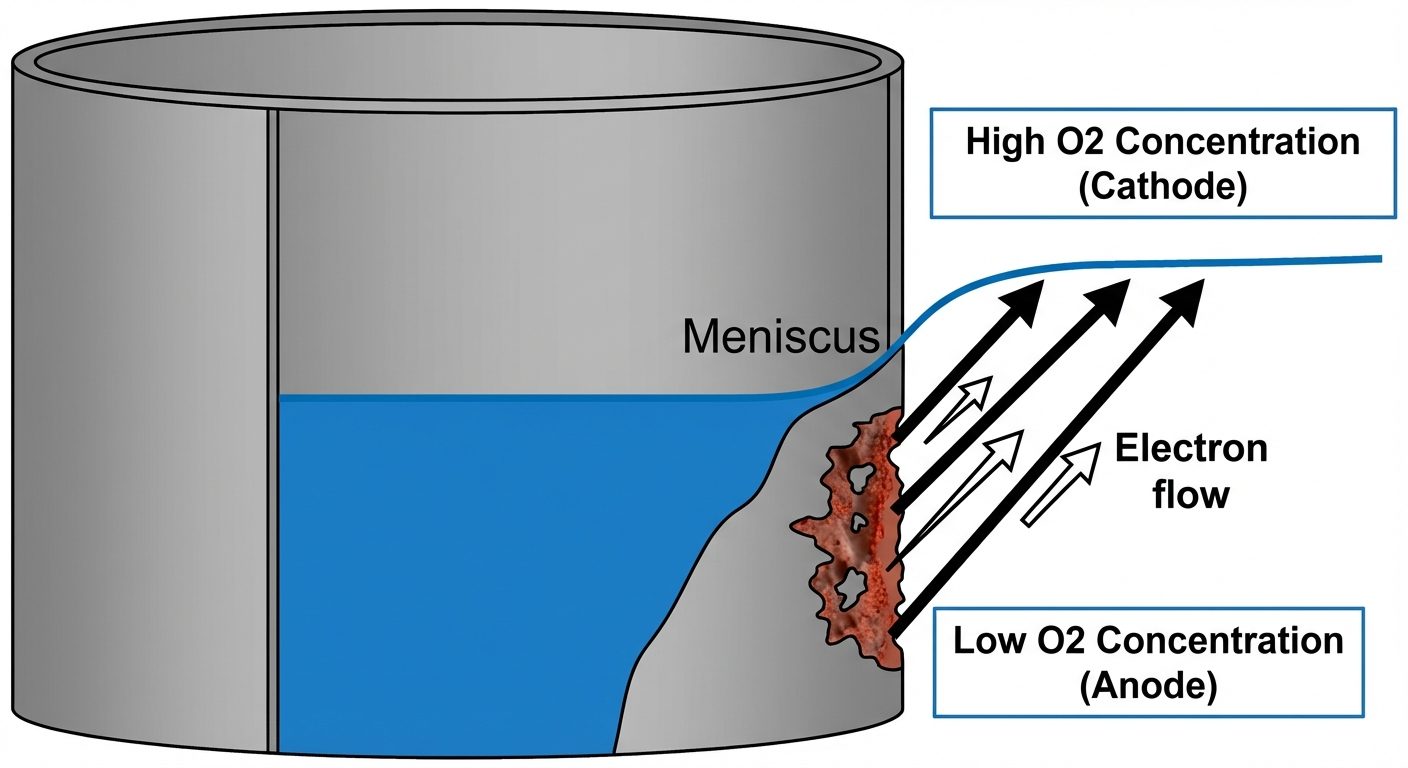
Water-line Corrosion
Occurs in steel tanks or ships partially filled with water.
- Observation: Corrosion occurs just below the water meniscus (water line).
- Reason:
- The area at the water line is well-aerated (Cathode).
- The area below the water line is poorly aerated (Anode).
- Outcome: A distinct groove of corrosion forms below the water level.
Pitting Corrosion
A localized form of corrosion that produces cavities or holes in the material.
- Mechanism: It starts at a break in the protective oxide layer. The small exposed area becomes the Anode (low oxygen availability inside the pit), while the large surrounding surface remains Cathodic (high oxygen).
- Danger: It is autocatalytic; the pit grows deeper rapidly while the surface looks relatively undamaged.
- Causes: Stagnant water, chloride ions (), surface scratches.
6. Other Types of Corrosion
Intergranular Corrosion
- Location: Occurs along the grain boundaries of a metal alloy rather than the grain interior.
- Common Scenario: Stainless Steel exposed to high temperatures (e.g., welding).
- Mechanism (Weld Decay): At high temps, Chromium combines with Carbon to form Chromium Carbide precipitates at the grain boundaries. This creates a chromium-depleted zone adjacent to the boundary, which becomes Anodic relative to the grain body.
- Result: The alloy disintegrates into grains.
Soil Corrosion
- Context: Affects underground pipelines, cables, and structures.
- Factors:
- Acidity: Acidic soils accelerate corrosion.
- Moisture: Acts as the electrolyte.
- Conductivity: Salts in soil increase conductivity and corrosion rate.
- Microbial: Sulphate-reducing bacteria in anaerobic soils can induce severe corrosion.
7. Factors Affecting Corrosion
Nature of the Metal
- Position in Galvanic Series: Metals higher in the series (more anodic) corrode faster. The greater the potential difference between two metals, the faster the corrosion.
- Relative Areas of Anode and Cathode:
- Small Anode + Large Cathode = Rapid Corrosion (Current density at anode is extremely high).
- Large Anode + Small Cathode = Slow/Negligible Corrosion.
- Purity: Pure metals resist corrosion better. Impurities usually act as small cathodic sites, setting up micro-galvanic cells.
- Physical State: Stressed or bent parts of a metal act as anodes (high energy) and corrode relative to unstressed parts.
Nature of the Environment
- Temperature: Rate of corrosion increases with temperature (reaction kinetics increase).
- Humidity: Higher humidity provides the necessary electrolyte layer (especially critical above "Critical Humidity").
- pH: Acidic pH generally increases corrosion (hydrogen evolution).
- Conductivity: High conductivity (e.g., seawater) increases the flow of corrosion current.
8. Protection of Corrosion
Corrosion control falls into three categories: Material Selection/Design, Cathodic Protection, and Surface Coatings.
A. Protection by Surface Coating
Isolates the metal from the environment.
- Metallic Coatings: Galvanizing (Zinc), Tinning (Tin).
- Organic Coatings: Paints, varnishes, lacquers.
- Inorganic Coatings: Phosphate, chromate, and ceramic coatings.
B. Ceramic Coating
Ceramic coatings are inorganic, non-metallic coatings used for resistance against high temperatures and aggressive chemicals.
- Materials Used: Alumina (), Zirconia (), Silica.
- Process: Applied via thermal spraying, sol-gel process, or chemical vapor deposition (CVD).
- Advantages:
- Excellent thermal barrier (protects against heat).
- Chemically inert (prevents oxidation and acid attack).
- High hardness and wear resistance.
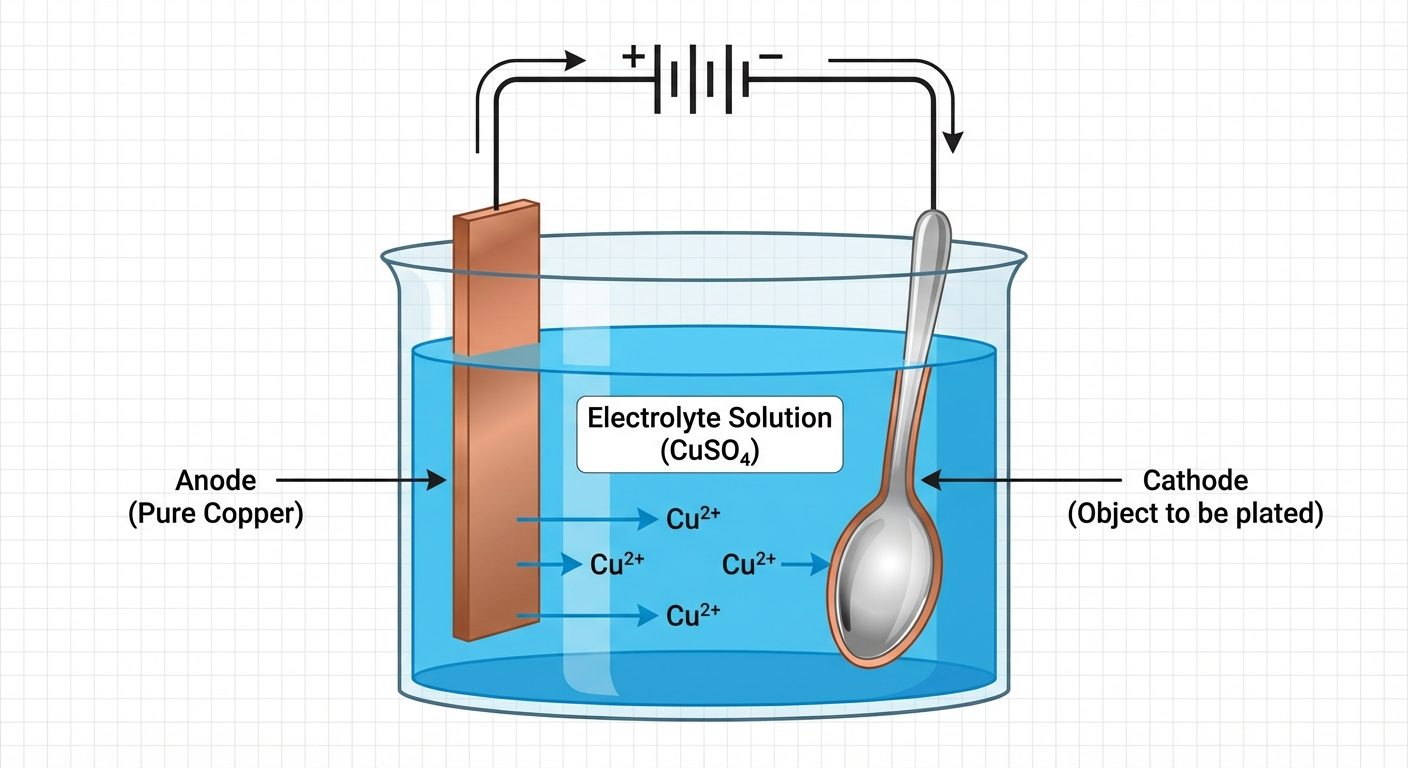
C. Electroplating Process
Electroplating is the process of coating a base metal with a layer of another metal using electrolysis.
Objectives: Decoration, corrosion protection, improving hardness.
Process Setup:
- Electrolytic Bath: A tank containing a salt solution of the coating metal.
- Anode: Made of the coating metal (e.g., Copper bar).
- Cathode: The base metal/object to be plated (e.g., Iron spoon).
- Power Source: DC current is applied.
Mechanism:
- At Anode (Oxidation): The coating metal dissolves into the electrolyte.
- At Cathode (Reduction): Metal ions from the solution deposit onto the object.
Key Conditions for Good Plating:
- Clean surface of the base metal.
- Optimum current density.
- Proper temperature and electrolyte concentration.