Unit 6 - Notes
Unit 6: Energy Sciences
1. Energy Sciences: Fuels and Classifications
Definition of Fuel
A fuel is a combustible substance containing carbon as the main constituent which, on proper burning, gives large amounts of heat that can be used economically for domestic and industrial purposes.
Classification of Fuels:
- Based on Occurrence:
- Primary (Natural) Fuels: Occur in nature (e.g., Wood, Coal, Petroleum, Natural Gas).
- Secondary (Artificial) Fuels: Derived from primary fuels (e.g., Coke, Gasoline, Coal gas).
- Based on Physical State:
- Solid Fuels: Wood, coal, coke.
- Liquid Fuels: Crude oil, petrol, diesel, alcohol.
- Gaseous Fuels: Natural gas, water gas, producer gas, hydrogen.
2. Calorific Value of Fuels
The total quantity of heat liberated when a unit mass (or volume) of a fuel is burnt completely.
Types of Calorific Value
- Higher Calorific Value (HCV) / Gross Calorific Value (GCV):
The total amount of heat produced when a unit quantity of fuel is completely burnt and the products of combustion are cooled down to room temperature ( or ). This includes the latent heat of condensation of steam produced. - Lower Calorific Value (LCV) / Net Calorific Value (NCV):
The net heat produced when a unit quantity of fuel is completely burnt and the products of combustion are permitted to escape.- Formula:
Determination of Calorific Value: Bomb Calorimeter
Used for determining the calorific value of solid and non-volatile liquid fuels.
- Principle: A known mass of fuel is burnt in a sealed steel vessel (bomb) filled with oxygen. The heat produced is absorbed by a known mass of water surrounding the bomb.
- Construction: Consists of a strong stainless steel bomb (to withstand high pressure), a copper calorimeter vessel, a water jacket (to minimize heat loss), a stirrer, and a Beckman thermometer.
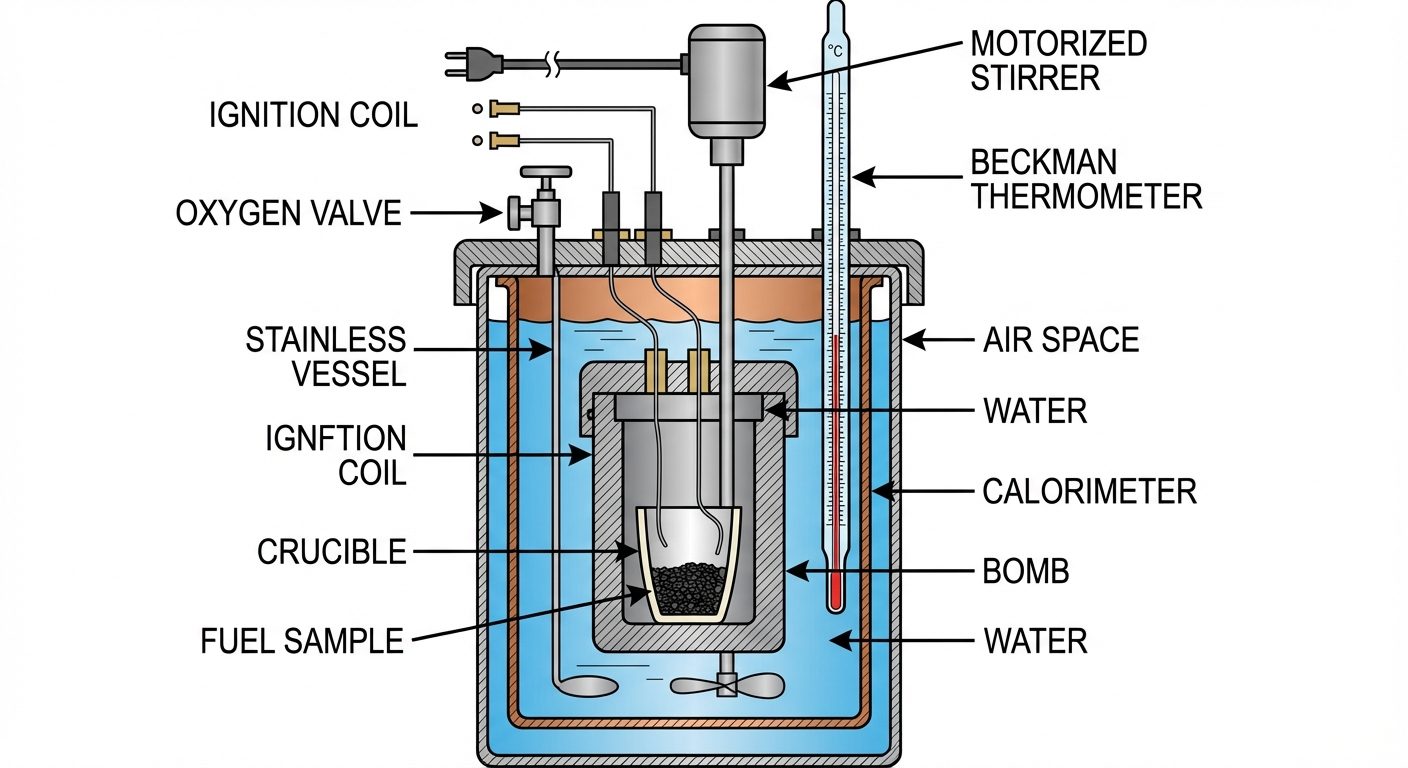
- Calculation:
- = Mass of water in calorimeter
- = Water equivalent of calorimeter
- = Initial temperature
- = Final temperature
- = Mass of fuel
3. Analysis of Coal
To assess the quality of coal, two types of analysis are performed:
A. Proximate Analysis (Physical Analysis)
Determines the behavior of coal when heated. It involves the determination of:
- Moisture: Loss in weight when coal is heated at . High moisture lowers calorific value.
- Volatile Matter: Loss in weight when moisture-free coal is heated at without air. High volatile matter causes long flames and smoke.
- Ash: Residue remaining after complete combustion at . Ash reduces calorific value and causes clinkering.
- Fixed Carbon: Calculated as . Higher fixed carbon implies higher calorific value.
B. Ultimate Analysis (Elemental Analysis)
Involves the determination of element percentages:
- Carbon & Hydrogen: Determined by combustion train method. Higher C and H increase quality.
- Nitrogen: Kjeldahl's method. Undesirable as it forms NOx.
- Sulfur: Eschka's method. Undesirable ( causes corrosion/pollution).
- Oxygen: Calculated by difference. Lower oxygen is preferred as it holds moisture.
4. Electrochemical Energy Systems: Batteries
Classification
- Primary Cells: Non-rechargeable. Cell reaction is irreversible. Example: Dry cell, Lithium primary cell.
- Secondary Batteries: Rechargeable. Cell reaction is reversible (chemical energy electrical energy). Example: Lead-acid, Li-ion.
Important Battery Technologies
1. Lead Storage Battery (Lead-Acid)
- Anode: Spongy Lead ().
- Cathode: Lead Dioxide ().
- Electrolyte: Dilute Sulfuric Acid (, approx 38%).
- Discharge Reaction:
- Anode:
- Cathode:
- Applications: Automobiles, UPS systems.
2. Nickel-Cadmium (Ni-Cd) Battery
- Anode: Cadmium ().
- Cathode: Nickel oxyhydroxide ().
- Electrolyte: Potassium Hydroxide ().
- Pros: Long shelf life, operates at low temps.
- Cons: "Memory effect" (reduced capacity if not fully discharged), Cadmium toxicity.
3. Nickel-Metal Hydride (Ni-MH) Battery
- Anode: Metal Hydride (, hydrogen-absorbing alloy).
- Cathode: Nickel oxyhydroxide ().
- Significance: Higher energy density than Ni-Cd, no toxic cadmium, less memory effect. Used in hybrid vehicles and consumer electronics.
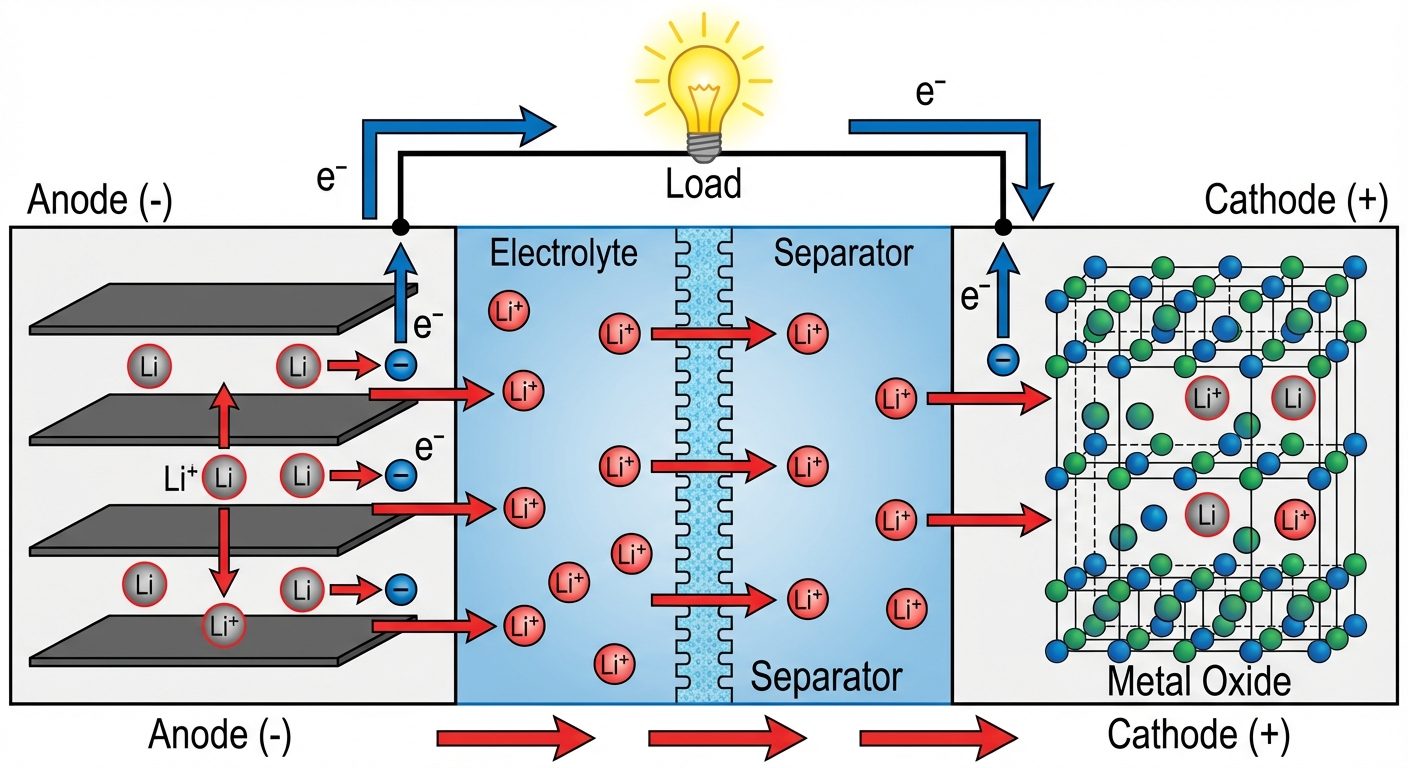
4. Lithium-Ion Battery (Li-ion)
Currently the most popular for portable electronics. Works on the principle of Intercalation (insertion of ions into the crystal lattice).
- Anode: Graphite (carbon layers).
- Cathode: Lithium metal oxide (e.g., , ).
- Electrolyte: Lithium salt in organic solvent ().
- Mechanism: During discharge, ions move from anode to cathode through the electrolyte. During charging, they move back.
5. Lithium-Air Battery (Li-Air)
- Concept: Uses oxidation of lithium at the anode and reduction of oxygen (from air) at the cathode.
- Potential: Extremely high theoretical energy density (comparable to gasoline).
- Challenge: Stability of electrolyte and cathode clogging by reaction products.
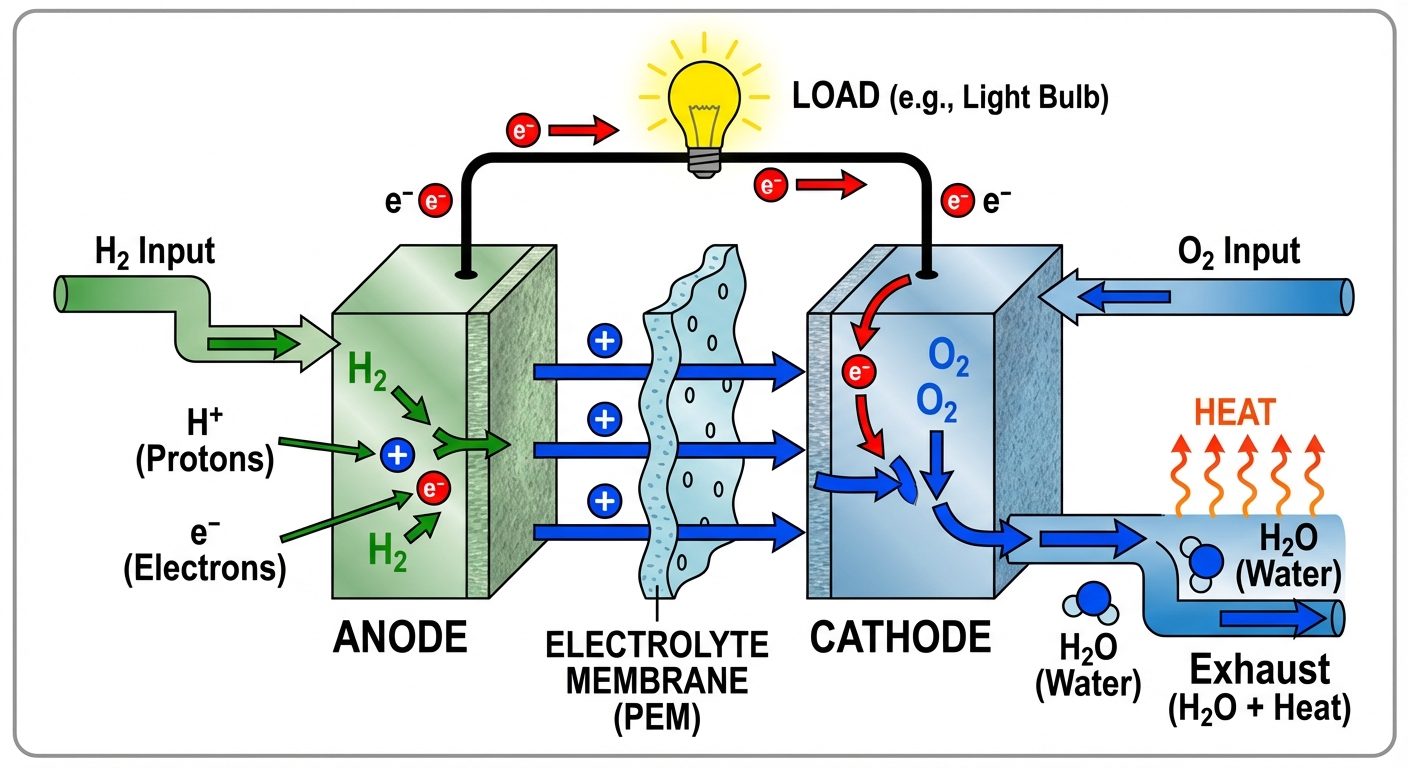
5. Fuel Cells
A galvanic cell that converts chemical energy of a fuel (hydrogen, methanol) directly into electrical energy without combustion. Reactants are supplied continuously.
Hydrogen-Oxygen () Fuel Cell
- Anode: Porous carbon containing Pt/Pd catalyst.
- Cathode: Porous carbon containing Pt/Pd catalyst.
- Electrolyte: KOH solution or Polymer Electrolyte Membrane (PEM).
- Reactions:
- Anode:
- Cathode:
- Overall:
Significance
- Advantages: High efficiency (60-70%), pollution-free (water is the byproduct), silent operation.
- Disadvantages: High cost of catalysts (Platinum), storage and transport of Hydrogen is difficult.
- Applications: Spacecraft (water produced is used for drinking), electric vehicles (FCEVs).
6. Hydrogen Energy
Hydrogen is considered the "Fuel of the Future" due to its high calorific value (150 kJ/g) and clean combustion.
Production
- Steam Methane Reforming (SMR): . (Most common).
- Electrolysis of Water: Splitting water using electricity (). Green Hydrogen if electricity is renewable.
Storage
- Compressed Gas: High-pressure tanks (350-700 bar).
- Liquid Hydrogen: Cryogenic tanks at .
- Solid State: Metal hydrides (MgH2) adsorb hydrogen (safest method).
Safety Aspects
- Hydrogen has a wide flammability range (4-75% in air).
- Colorless and odorless flame (hard to detect).
- Low ignition energy required.
7. Nuclear Energy
Principles
- Nuclear Fission: Splitting a heavy nucleus () into lighter nuclei by neutron bombardment, releasing massive energy.
- Reaction: .
- Nuclear Fusion: Combining light nuclei () to form a heavier nucleus. Occurs in the sun. Higher energy yield but requires extreme temperatures.
Sustainable Energy Production
- Nuclear power provides baseload electricity with near-zero carbon emissions.
- Advanced reactors (Generation IV) aim to use nuclear waste as fuel, reducing radiotoxicity and increasing sustainability.
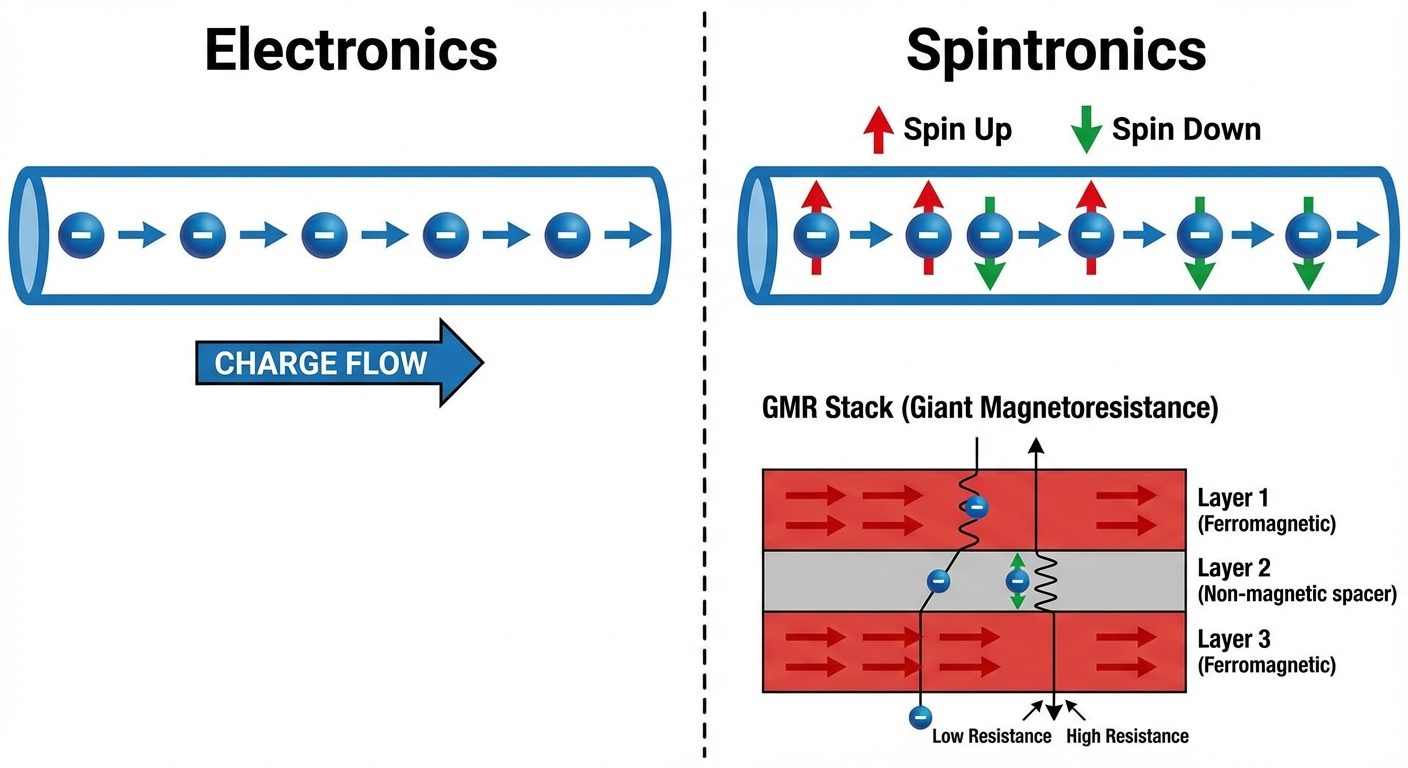
8. Spintronics (Spin Transport Electronics)
Definition
A field of electronics that exploits the intrinsic spin of the electron and its associated magnetic moment, in addition to its fundamental electronic charge.
Principle: Giant Magnetoresistance (GMR)
A quantum mechanical effect observed in thin-film structures composed of alternating ferromagnetic and non-magnetic conductive layers. The electrical resistance drops significantly when the magnetic fields of the layers are aligned (parallel) compared to when they are anti-parallel.
Engineering Applications
- Data Storage: Read heads in Hard Disk Drives (HDD) use GMR sensors to read high-density data.
- MRAM (Magnetoresistive Random Access Memory): Non-volatile memory that uses magnetic states to store bits. It is faster than Flash and denser than SRAM.
- Quantum Computing: Electron spin is used as a Qubit (quantum bit) for processing information.