Unit 4 - Notes
Unit 4: Electrochemistry
1. Introduction to Electrolytes and Conductance
1.1 Electrolytes
Substances that conduct electricity in their molten state or aqueous solution due to the movement of ions are called electrolytes.
- Strong Electrolytes: Completely dissociate into ions (e.g., HCl, NaOH, NaCl). High conductivity.
- Weak Electrolytes: Partially dissociate (e.g., CH₃COOH, NH₄OH). Low conductivity.
1.2 Concepts of Conductance
In metallic conductors, current is carried by electrons. In electrolytic conductors, current is carried by ions.
A. Resistance ()
- Obstruction to the flow of current.
- Unit: Ohm ()
- Formula:
- = distance between electrodes (cm)
- = cross-sectional area of electrodes (cm²)
- = Specific resistance (Resistivity)
B. Conductance ()
- Reciprocal of resistance.
- Unit: Siemens () or Ohm⁻¹ () or mho.
- Formula:
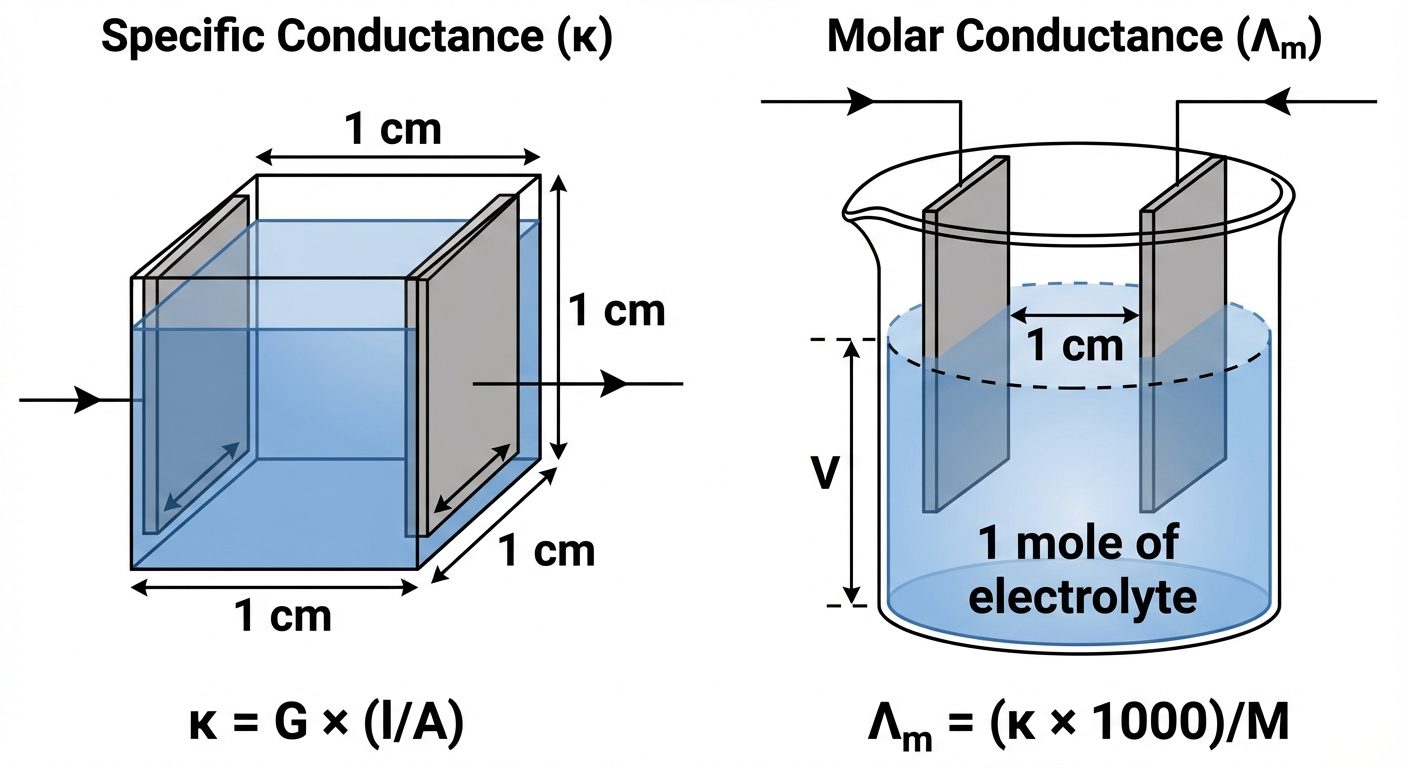
C. Specific Conductance ( - Kappa)
- Conductance of a solution enclosed between two electrodes of 1 cm² area separated by 1 cm distance (i.e., conductance of 1 cm³ of solution).
- Formula:
- Unit: or
D. Molar Conductance ()
- Conductance of all ions produced by dissolving 1 mole of electrolyte in cm³ of solution.
- Formula:
- = Molarity (mol/L)
- Unit:
2. Cell Constant and Its Determination
Definition
The ratio of the distance between the electrodes () to the area of cross-section () of the electrodes is a constant for a specific conductivity cell.
- Unit: or
Determination (Experimental)
Since measuring and physically is difficult due to irregular electrode surfaces, the cell constant is determined indirectly:
- A standard solution of KCl (whose specific conductance is known accurately at various temperatures) is used.
- The resistance () of this KCl solution is measured using a Wheatstone bridge circuit.
- Calculation:
Numerical Example 1 (Conductance)
Problem: A conductivity cell filled with 0.1 M KCl solution has a resistance of 100 . If the specific conductance of 0.1 M KCl is 1.29 , calculate the cell constant. If the same cell is filled with 0.02 M electrolyte solution offering a resistance of 520 , calculate the Molar Conductance of the 0.02 M solution.
Solution:
-
Calculate Cell Constant ():
-
Calculate of 0.02 M solution:
Convert to : -
Calculate Molar Conductance ():
3. Electrochemical Cells
An electrochemical cell is a device capable of either generating electrical energy from chemical reactions or using electrical energy to cause chemical reactions.
3.1 Types of Cells
| Feature | Electrolytic Cell | Galvanic (Voltaic) Cell |
|---|---|---|
| Energy Conversion | Electrical Energy Chemical Energy | Chemical Energy Electrical Energy |
| Spontaneity | Non-spontaneous () | Spontaneous () |
| Anode Charge | Positive (+) | Negative (-) |
| Cathode Charge | Negative (-) | Positive (+) |
| Electron Flow | Into cell at cathode, out at anode | Out of anode, into cathode |
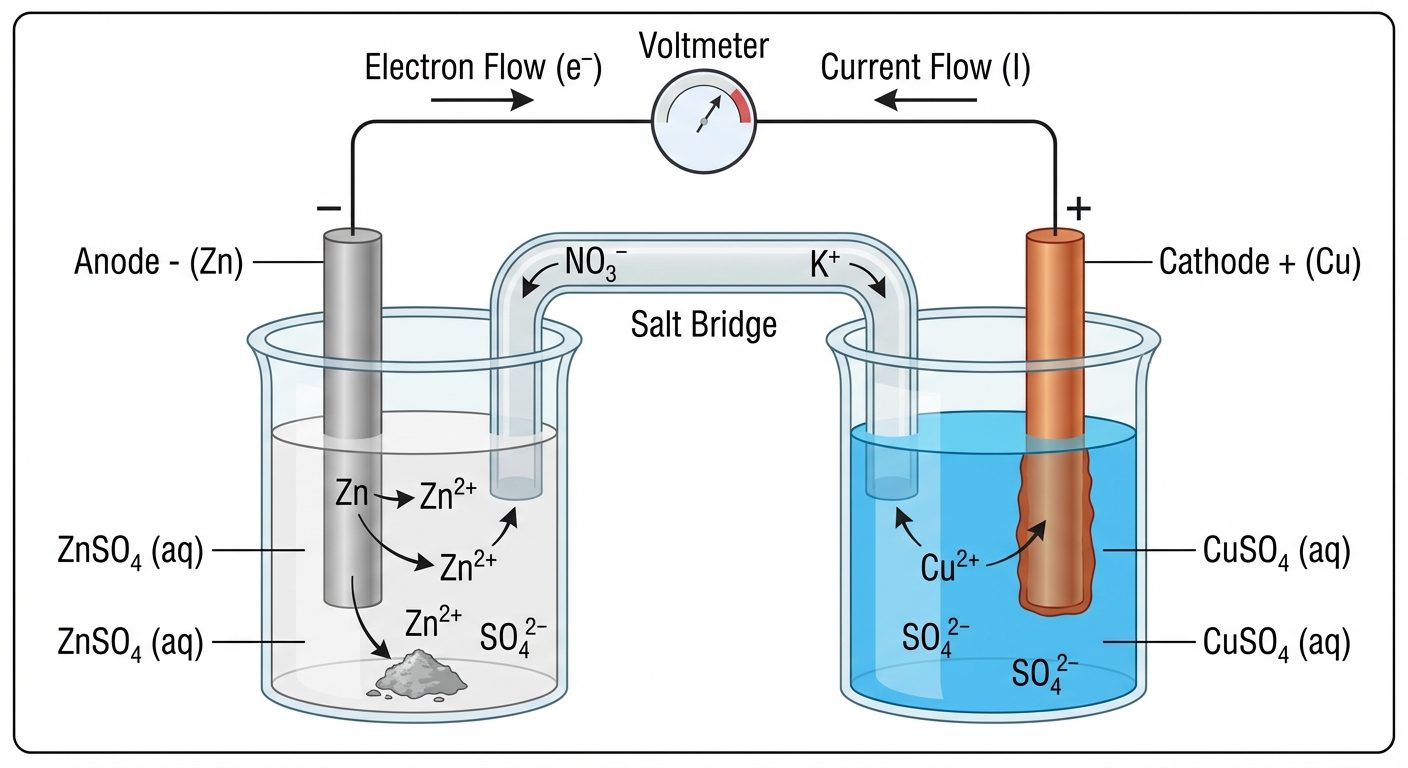
3.2 Galvanic Cell Structure (Daniel Cell)
A classic example consisting of Zinc and Copper half-cells.
- Anode (Oxidation): Zinc rod in ZnSO₄ solution.
- Cathode (Reduction): Copper rod in CuSO₄ solution.
- Salt Bridge: A U-tube containing inert electrolyte (KCl/KNO₃) in agar-agar jelly. It maintains electrical neutrality and completes the circuit.
4. Electrode Potentials
4.1 Origin of Single Electrode Potential
When a metal () is placed in a solution of its own ions (), two opposing tendencies operate:
- Solution Pressure (): Tendency of metal atoms to lose electrons and pass into solution as cations (Oxidation).
- Osmotic Pressure (): Tendency of metal ions from solution to gain electrons and deposit on the metal (Reduction).
- If : Oxidation occurs (Anode). The rod becomes negatively charged.
- If : Reduction occurs (Cathode). The rod becomes positively charged.
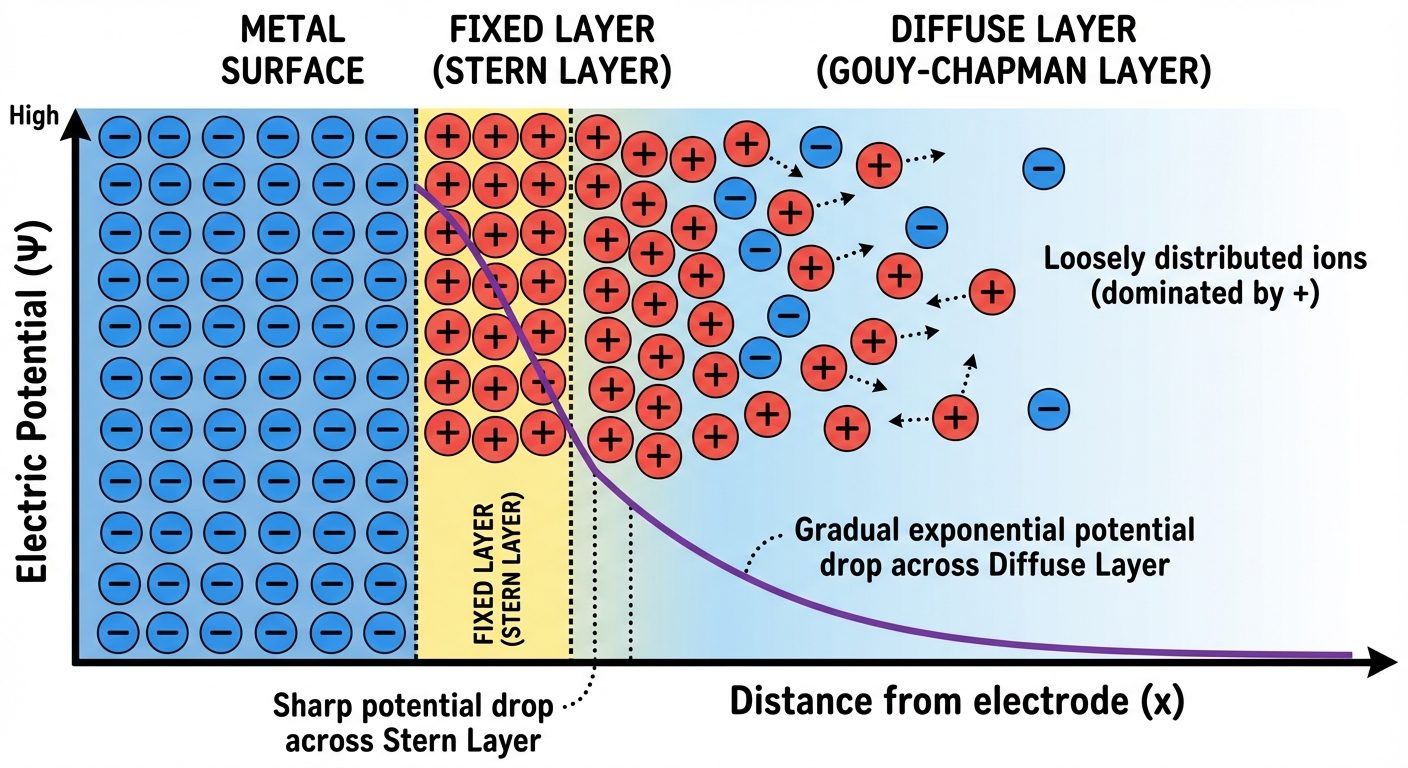
4.2 Helmholtz Double Layer (HDL)
The separation of charges at the interface between the electrode and the electrolyte creates a potential difference.
- Fixed Layer: A layer of ions adheres directly to the electrode surface (e.g., if the rod is negative, positive ions adsorb).
- Diffuse Layer: A layer of ions of opposite charge distributed in the solution near the fixed layer.
This arrangement acts like a parallel plate capacitor, generating the electrode potential.
4.3 Electrochemical Series
Arrangement of elements in increasing order of their Standard Reduction Potentials ().
- Top (Negative ): Strong reducing agents (Li, K, Na, Zn). Active metals.
- Hydrogen: V (Reference).
- Bottom (Positive ): Strong oxidizing agents (Cu, Ag, Au, F₂). Noble metals.
5. Cell EMF and Nernst Equation
5.1 Cell EMF
The Electromotive Force (EMF) is the potential difference between the two electrodes when no current flows.
5.2 Nernst Equation
Dependence of electrode potential on concentration and temperature.
For a half-cell reaction:
Since (solid) = 1, and converting to at 298 K ():
For a full cell:
Numerical Example 2 (Nernst Equation)
Problem: Calculate the EMF of the cell at :
Given: , .
Solution:
-
Cell Reaction:
Here, electrons involved. -
Calculate :
-
Apply Nernst Equation:
6. Thermodynamics of Electrochemical Cells
The electrical work done by a cell is equal to the decrease in Gibbs Free Energy ().
Key Relationships
-
Gibbs Free Energy:
- For standard conditions:
- If is positive, is negative (Spontaneous).
-
Enthalpy Change ():
-
Entropy Change ():
- is the Temperature Coefficient of the cell.
7. Reversible and Irreversible Cells
7.1 Reversible Cells
A cell is reversible if it satisfies thermodynamic conditions of reversibility:
- If an external EMF exactly equal to the cell EMF is applied, no current flows and no chemical reaction occurs.
- If the external EMF is infinitesimally smaller than the cell EMF, current flows from the cell (Discharging).
- If the external EMF is infinitesimally larger than the cell EMF, current flows into the cell and the cell reaction is reversed (Charging).
- Example: Daniel Cell (Zn/Cu).
7.2 Irreversible Cells
Cells that do not obey the reversibility conditions. Even if the current direction is reversed, the chemical changes are not completely reversed (often due to gas evolution or side reactions).
- Example: Zinc-Silver cell involving H₂SO₄ where Hydrogen gas escapes.