Unit 3 - Notes
Unit 3: Nanomaterials
1. Introduction to Nanomaterials
Nanomaterials are defined as materials with at least one external dimension in the size range from approximately 1 to 100 nanometers (). At this scale, materials exhibit properties (physical, chemical, and biological) that differ significantly from those of bulk materials.
Key Concept: The transition from bulk to nano leads to a drastic increase in the Surface Area to Volume Ratio and the dominance of Quantum Effects.
1.1 Classification Based on Dimensionality
Nanomaterials are classified based on the number of dimensions that are outside the nanoscale range ().
- Zero-Dimensional (0D): All three dimensions () are within the nanoscale (1-100 nm).
- Examples: Quantum dots, nanoparticles, fullerenes.
- One-Dimensional (1D): Two dimensions are in the nanoscale; one dimension is outside (macroscopic).
- Examples: Nanowires, nanotubes, nanorods.
- Two-Dimensional (2D): One dimension is in the nanoscale; two dimensions are outside.
- Examples: Thin films, graphene, nanocoatings.
- Three-Dimensional (3D): No dimensions are in the nanoscale (all ), but the material possesses a nanocrystalline structure or bulk structure made of nanocomponents.
- Examples: Bulk nanocomposites, polycrystals with grains in nanosize.
[Image generation failed: A detailed schematic diagram illustrating the clas...]
2. Properties of Nanomaterials (Size-Dependent Effects)
2.1 Electronic Properties
- Quantum Confinement: In bulk materials, energy bands (valence and conduction) are continuous. In nanomaterials, these bands become discrete energy levels.
- Band Gap Expansion: As size decreases, the band gap energy increases. Conductors may become semiconductors or insulators at the nanoscale.
2.2 Optical Properties
- Surface Plasmon Resonance (SPR): The oscillation of free electrons on the surface of metallic nanoparticles in resonance with incident light causes unique colors.
- Example: Gold (Au) bulk is yellow, but Au nanoparticles appear ruby red or purple depending on size.
- Blue Shift: Due to the increased band gap, absorption and emission spectra shift towards shorter wavelengths (blue end).
2.3 Mechanical Properties
- Increased Strength and Hardness: The Hall-Petch relation suggests that strength increases as grain size decreases (fewer dislocations).
- Ductility: Some ceramics, brittle in bulk, become ductile at the nanoscale due to grain boundary sliding.
2.4 Thermal Properties
- Melting Point Depression: The melting point of nanoparticles is significantly lower than that of the bulk material because surface atoms (which are less stable) require less energy to move.
- Thermal Conductivity: Nanowires may show reduced thermal conductivity due to phonon scattering at boundaries (useful for thermoelectrics), while Carbon Nanotubes (CNTs) show exceptionally high conductivity.
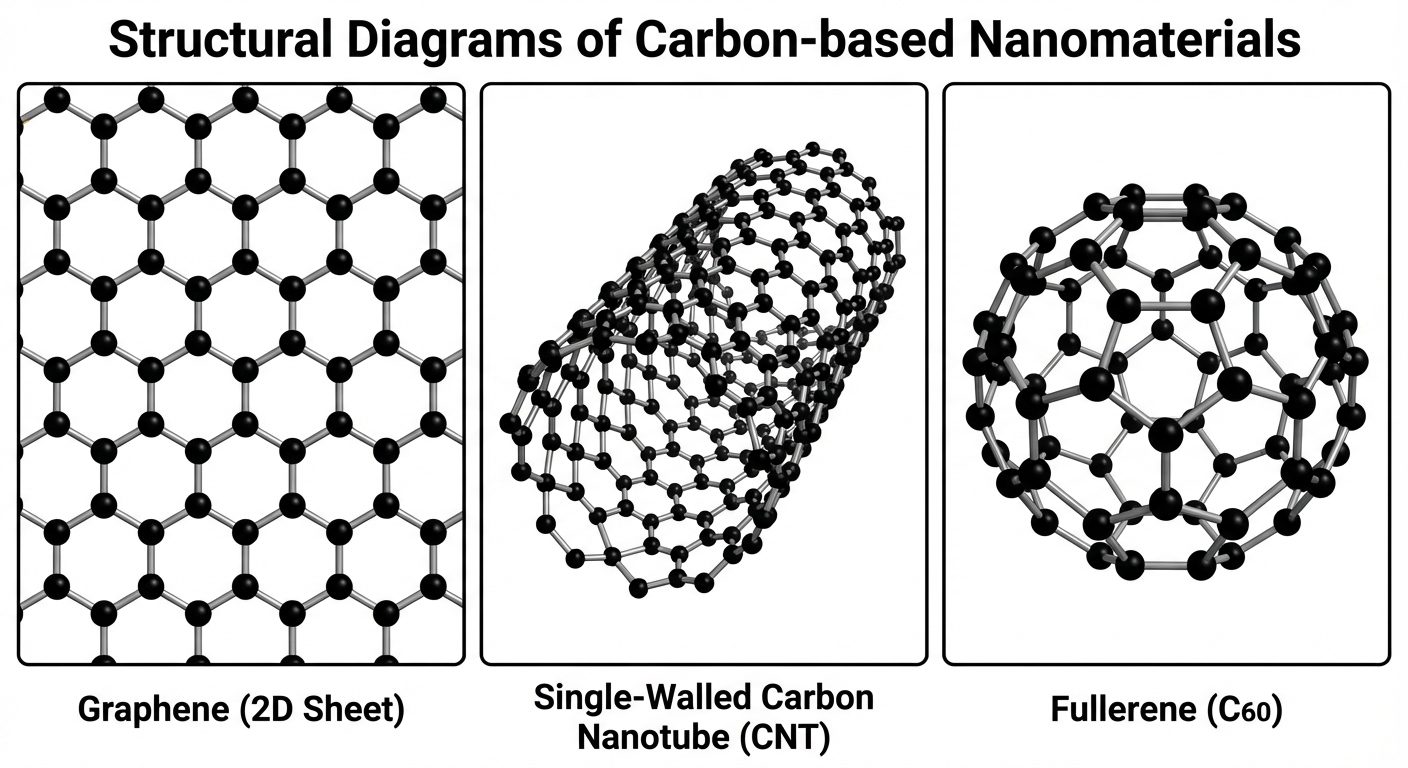
3. Carbon-Based Nanomaterials
3.1 Fullerenes (Buckyballs)
- Structure: Spherical cage-like molecules composed of hybridized carbon atoms arranged in hexagons and pentagons (similar to a soccer ball).
- C60 (Buckminsterfullerene): The most common form, containing 60 carbon atoms with 20 hexagons and 12 pentagons.
- Properties: High chemical stability, electron-accepting ability, soluble in organic solvents.
- Applications: Drug delivery vehicles, antioxidants, organic photovoltaics.
3.2 Carbon Nanotubes (CNTs)
- Structure: Sheets of graphene rolled into seamless cylinders.
- Single-Walled CNTs (SWCNTs): One layer of graphene rolled up (Diameter: ~1 nm).
- Multi-Walled CNTs (MWCNTs): Concentric cylinders of graphene sheets (Diameter: 10-100 nm).
- Properties:
- Mechanical: Tensile strength 100x stronger than steel.
- Electrical: Can be metallic or semiconducting depending on chirality (rolling angle).
- Thermal: Excellent heat conductors.
3.3 Graphene
- Structure: A single planar sheet of bonded carbon atoms arranged in a honeycomb lattice. It is the basic building block for graphite, CNTs, and fullerenes.
- Properties: Zero-bandgap semiconductor, high electron mobility, transparent, strongest material ever tested.
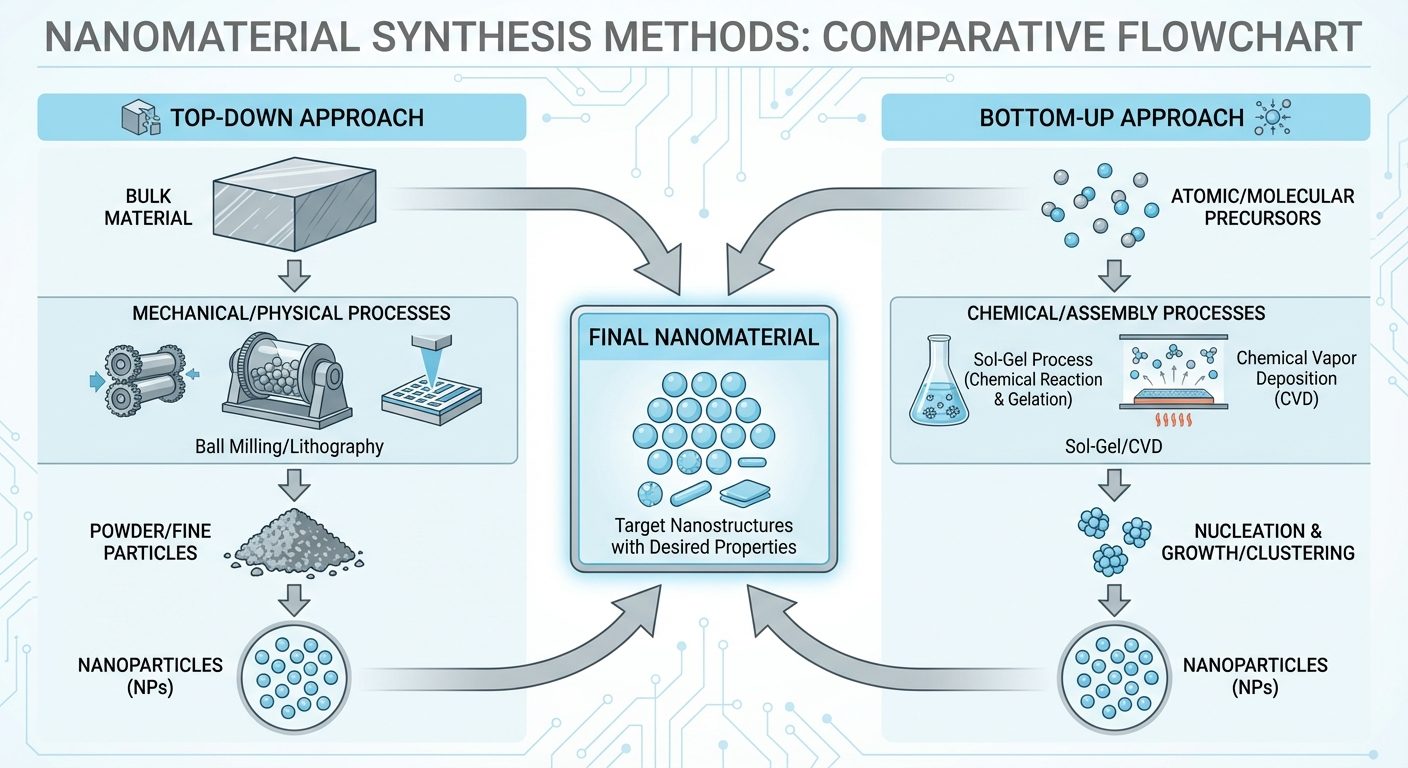
4. Synthesis of Nanomaterials
4.1 Top-Down Approach
Breaking down bulk material into nano-sized particles.
- Ball Milling: Mechanical crushing using hard balls in a rotating container.
- Pros: Simple, scalable.
- Cons: Irregular shapes, contamination from milling media.
- Lithography: Using light or electron beams to etch patterns onto a substrate (used in chip manufacturing).
4.2 Bottom-Up Approach
Assembling atoms or molecules to form nanostructures.
- Sol-Gel Method: Transition from a liquid "sol" (colloidal suspension) to a solid "gel" network. Excellent for metal oxides.
- Chemical Vapor Deposition (CVD): Volatile precursors react on a heated substrate to deposit a solid thin film or nanotube.
- Self-Assembly: Spontaneous organization of molecules into stable structures.
5. Applications of Nanomaterials
5.1 Catalysis
- High surface-to-volume ratio provides more active sites for reactions.
- Example: Platinum nanoparticles in catalytic converters for automobiles.
5.2 Electronics and Telecommunication
- Transistors: Nanoscale transistors (FETs) allow for smaller, faster processors (Moore’s Law).
- Displays: Quantum dots used in QLED TVs for vibrant colors.
- Sensors: CNT-based sensors for detecting gas leaks with high sensitivity.
5.3 Biomedical and Pharmaceutical
- Drug Delivery: Nanocarriers (liposomes, dendrimers) target specific cells (e.g., cancer), reducing side effects.
- Imaging: Quantum dots serve as contrast agents for MRI and fluorescence imaging.
5.4 Energy Technologies
- Batteries: Silicon nanowires in anodes allow for expansion, improving capacity in Li-ion batteries.
- Solar Cells: Nanoparticles increase light absorption efficiency in photovoltaic cells.
- Supercapacitors: Graphene and CNTs provide massive surface area for charge storage.
6. Composites
6.1 Fundamental Concepts
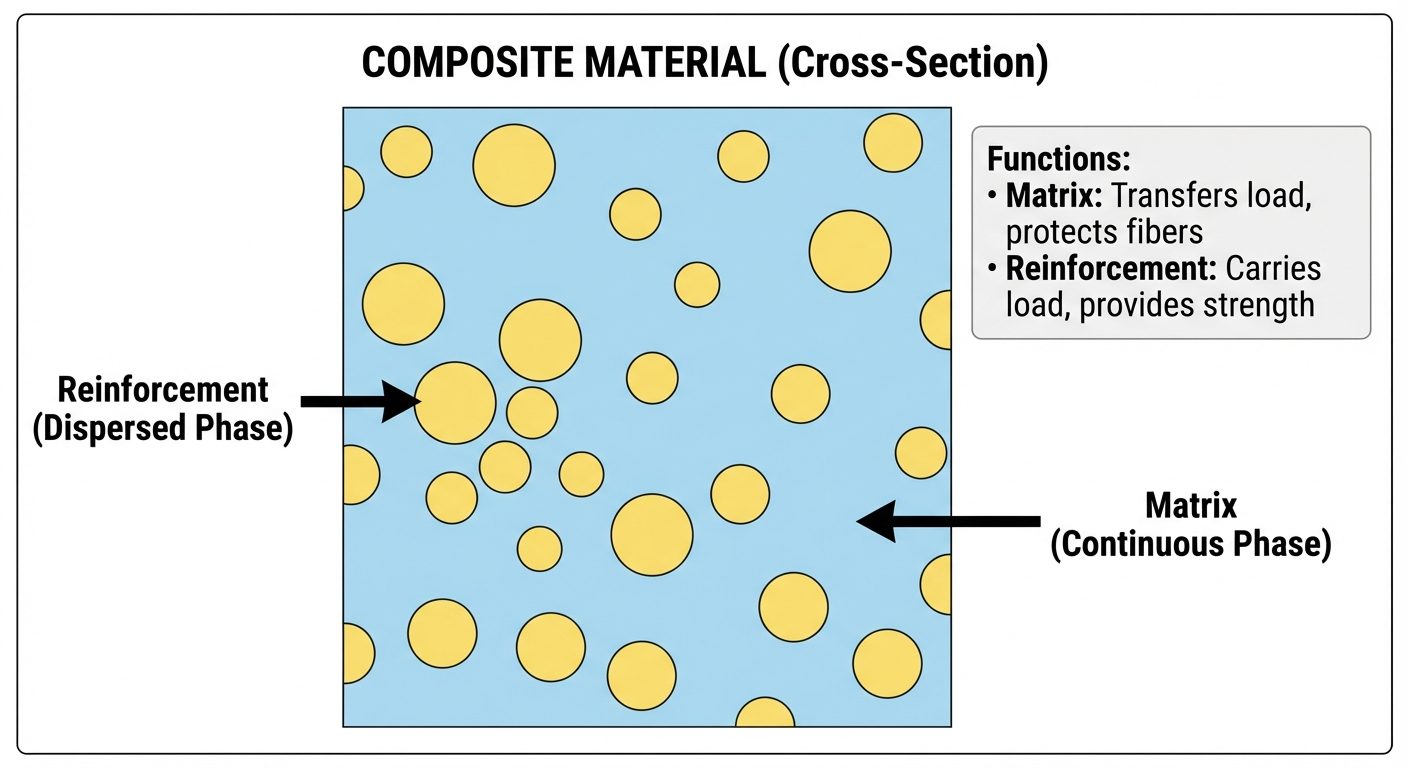
A Composite Material is a multiphase material formed from a combination of two or more chemically distinct materials to obtain properties superior to those of the individual components.
6.2 Components
- Matrix (Continuous Phase):
- Binds the reinforcement together.
- Transfers load to the reinforcement.
- Protects reinforcement from environmental damage.
- Types: Polymer, Metal, Ceramic.
- Reinforcement (Dispersed Phase):
- The primary load-bearing component.
- Provides strength, stiffness, and thermal resistance.
- Types: Fibers, particulates, flakes.
7. Classification of Composites
7.1 Based on Matrix Type
- Polymer Matrix Composites (PMCs): Most common (e.g., Carbon Fiber Reinforced Polymer). Low temperature applications.
- Metal Matrix Composites (MMCs): Metal matrix (Al, Ti) reinforced with ceramics. High wear resistance.
- Ceramic Matrix Composites (CMCs): Ceramic matrix reinforced with ceramic fibers to improve fracture toughness.
7.2 Based on Reinforcement Type
A. Particulate Composites
- Reinforcement consists of particles (spheres, platelets, irregular shapes).
- Dispersion-Strengthened: Very small particles () block dislocation movement (e.g., Thoria-dispersed Nickel).
- Large-Particle: Particles share the load (e.g., Concrete: gravel in cement; Cermets: WC in Cobalt).
B. Fiber-Reinforced Composites (FRCs)
- Reinforcement is in the form of fibers (high length-to-diameter ratio).
- Continuous (Aligned) Fibers: Fibers span the length of the composite. Anisotropic properties (strongest in fiber direction).
- Discontinuous (Short) Fibers: Chopped fibers. Can be aligned or randomly oriented.
C. Structural Composites
- Laminates: Stacked sheets (plies) with different fiber orientations to provide strength in multiple directions (e.g., Plywood).
- Sandwich Panels: Two strong outer sheets (face sheets) separated by a lightweight core (honeycomb or foam). High stiffness-to-weight ratio.
8. Engineering Applications of Composites
- Aerospace:
- Carbon/Epoxy composites used in wings and fuselage (Boeing 787, Airbus A350) to reduce weight and fuel consumption.
- Automotive:
- Glass fiber composites for body panels, bumpers, and drive shafts to improve fuel efficiency and crash safety.
- Construction:
- Concrete (aggregate + cement) is the most widely used particulate composite.
- Fiber-reinforced concrete for bridges and tunnels.
- Sports Equipment:
- Tennis rackets, golf clubs, and bicycle frames made of carbon fiber composites for high stiffness and low weight.
- Biomedical:
- Dental fillings (composite resins).
- Prosthetic limbs using carbon fiber.