Unit 2 - Notes
Unit 2: Polymers
1. Basics of Polymer Chemistry
Definitions
- Polymer: A large molecule (macromolecule) formed by the repeated linking of small structural units called monomers via covalent bonds.
- Monomer: A simple, low molecular weight molecule capable of combining with others to form a polymer.
- Polymerization: The chemical process of linking monomers together to form a polymer.
- Functionality: The number of bonding sites or reactive functional groups present in a monomer.
- Bifunctional: Forms linear polymers (e.g., Ethylene).
- Trifunctional/Polyfunctional: Forms branched or cross-linked polymers (e.g., Phenol).
2. Classification of Polymers
A. Based on Source
- Natural Polymers: Found in plants and animals (e.g., Starch, Cellulose, Proteins, Natural Rubber).
- Synthetic Polymers: Man-made in laboratories (e.g., Polyethylene, PVC, Nylon, Teflon).
- Semi-synthetic Polymers: Chemically modified natural polymers (e.g., Cellulose acetate, Rayon, Vulcanized rubber).
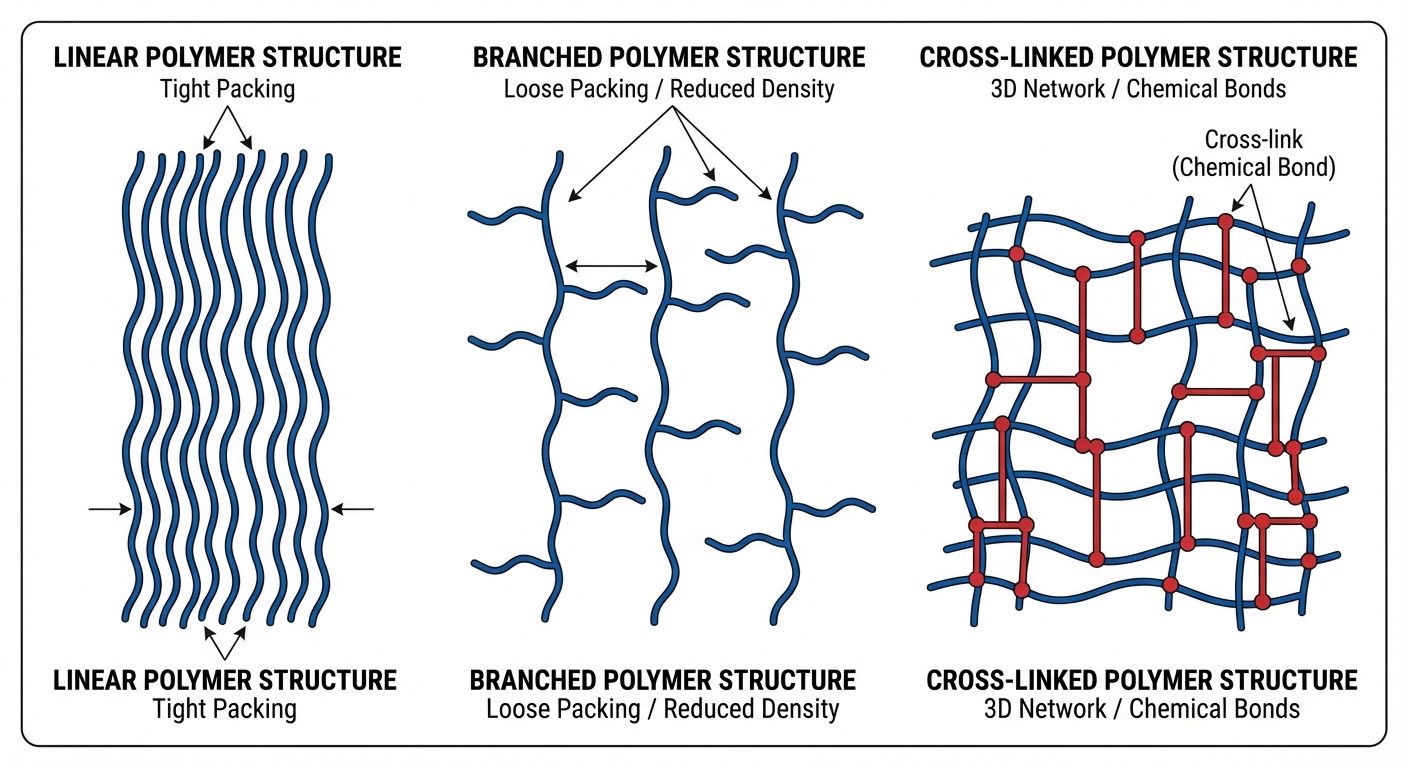
B. Based on Structure
- Linear Polymers: Long, continuous chains. High density and melting point due to close packing (e.g., HDPE, PVC).
- Branched Polymers: Chains with side branches. Lower density and melting point due to irregular packing (e.g., LDPE).
- Cross-linked (Network) Polymers: Chains linked by covalent bonds in a 3D network. Hard, rigid, and brittle (e.g., Bakelite, Melamine).
C. Based on Mode of Polymerization
- Addition (Chain Growth) Polymerization:
- Monomers add to the chain without eliminating by-products.
- Requires unsaturated monomers (containing double/triple bonds).
- Mechanism: Free radical, Cationic, or Anionic.
- Example: Ethylene Polyethylene.
- Condensation (Step Growth) Polymerization:
- Monomers combine with the elimination of small molecules (water, HCl, alcohol).
- Requires monomers with bifunctional or polyfunctional groups.
- Example: Hexamethylenediamine + Adipic acid Nylon 6,6 + .
D. Based on Molecular Forces
- Elastomers: Weak intermolecular forces (Van der Waals); elastic properties (e.g., Rubber).
- Fibers: Strong hydrogen bonds/dipole interactions; high tensile strength (e.g., Nylon, Polyester).
- Thermoplastics: Intermediate forces; soften on heating, harden on cooling; recyclable (e.g., PVC, Polyethylene).
- Thermosetting Plastics: Extensive cross-linking during molding; infusible and insoluble once set; cannot be remolded (e.g., Bakelite).
3. Degree of Polymerization (DP)
- Definition: The number of repeating units () in a polymer chain.
- Formula:
- Significance:
- Low DP: Oils or oligomers.
- High DP: Solid polymers with high mechanical strength.
- As DP increases, viscosity, boiling point, and mechanical strength increase up to a threshold.
4. Structure-Property Relationship
Molecular Shape
- Linear chains: Allow close packing, leading to high density, crystallinity, and tensile strength.
- Branched chains: Disrupt packing, lowering density and melting points.
Crystallinity
Polymers are rarely 100% crystalline; they are semi-crystalline (mix of amorphous and crystalline regions).
- Crystalline Regions: Ordered arrangement of chains (lamellae). Provides strength, stiffness, and chemical resistance.
- Amorphous Regions: Disordered/random arrangement. Provides flexibility and impact resistance.
- Factors aiding crystallinity: Linear structure, simple monomer structure, high intermolecular forces (H-bonding).
5. Glass Transition Temperature ()
Definition
The temperature at which a polymer transitions from a hard, glassy, brittle state to a soft, rubbery, flexible state.
- Below : Polymer is glassy/brittle (segmental motion is frozen).
- Above : Polymer is rubbery/flexible.
- Note: is for amorphous regions; Melting Point () is for crystalline regions.
Basic Factors Affecting
- Chain Flexibility: Rigid chains (aromatic rings in backbone) High . Flexible chains Low .
- Intermolecular Forces: Strong forces (H-bonding, polar groups) restrict motion High .
- Side Groups (Steric Hindrance): Bulky side groups hinder rotation High .
- Example: Polystyrene (bulky phenyl group, ) vs. Polyethylene (small H atom, ).
- Cross-linking: Restricts segmental motion Increases .
6. Structure, Synthesis, and Applications of Common Polymers
A. Elastomers (Synthetic Rubbers)
1. Styrene-Butadiene Rubber (SBR / Buna-S)
- Synthesis: Copolymerization of Styrene (25%) + 1,3-Butadiene (75%).
- Properties: High abrasion resistance, high load-bearing capacity.
- Applications: Automobile tires, footwear soles, conveyor belts.
2. Nitrile Rubber (NBR / Buna-N)
- Synthesis: Copolymerization of Acrylonitrile + 1,3-Butadiene.
- Properties: Excellent resistance to oils, fuels, and solvents due to polar CN groups.
- Applications: Oil seals, fuel tanks, hoses, gaskets.
B. Common Plastics
1. Polyvinyl Chloride (PVC)
- Monomer: Vinyl Chloride ().
- Structure: Linear chain with Chlorine pendant groups.
- Applications: Pipes, cable insulation, raincoats, flooring.
2. Teflon (Polytetrafluoroethylene - PTFE)
- Monomer: Tetrafluoroethylene ().
- Properties: Chemically inert, high thermal stability, low coefficient of friction, non-stick.
- Applications: Non-stick cookware coatings, bearings, gaskets, chemical storage liners.
3. PMMA (Polymethyl Methacrylate / Plexiglass)
- Properties: High optical transparency, weather resistance.
- Applications: Aircraft windows, lenses, automotive tail lights, skylights.
4. Kevlar (Aromatic Polyamide)
- Synthesis: Condensation of terephthaloyl chloride + p-phenylenediamine.
- Structure: Rigid aromatic backbone with inter-chain hydrogen bonds.
- Properties: Extremely high tensile strength, heat resistance, lightweight.
- Applications: Bulletproof vests, aerospace components, high-performance cables.
7. Biodegradable Polymers
Classification
- Natural: Starch, cellulose, collagen, chitosan.
- Synthetic: Polylactic acid (PLA), Polycaprolactone (PCL), Polyglycolic acid (PGA).
- Microbial: Polyhydroxyalkanoates (PHAs).
Methods of Degradation
- Hydrolysis: Breakdown of ester/amide bonds by water (common in PLA/PGA).
- Enzymatic Degradation: Microorganisms secrete enzymes (proteases, cellulases) to digest polymer chains.
- Photo-degradation: Light energy breaks polymer bonds.
Applications
- Medical: Dissolvable surgical sutures, drug delivery systems, bone pins.
- Packaging: Compostable bags, food containers.
- Agriculture: Mulch films that degrade into soil.
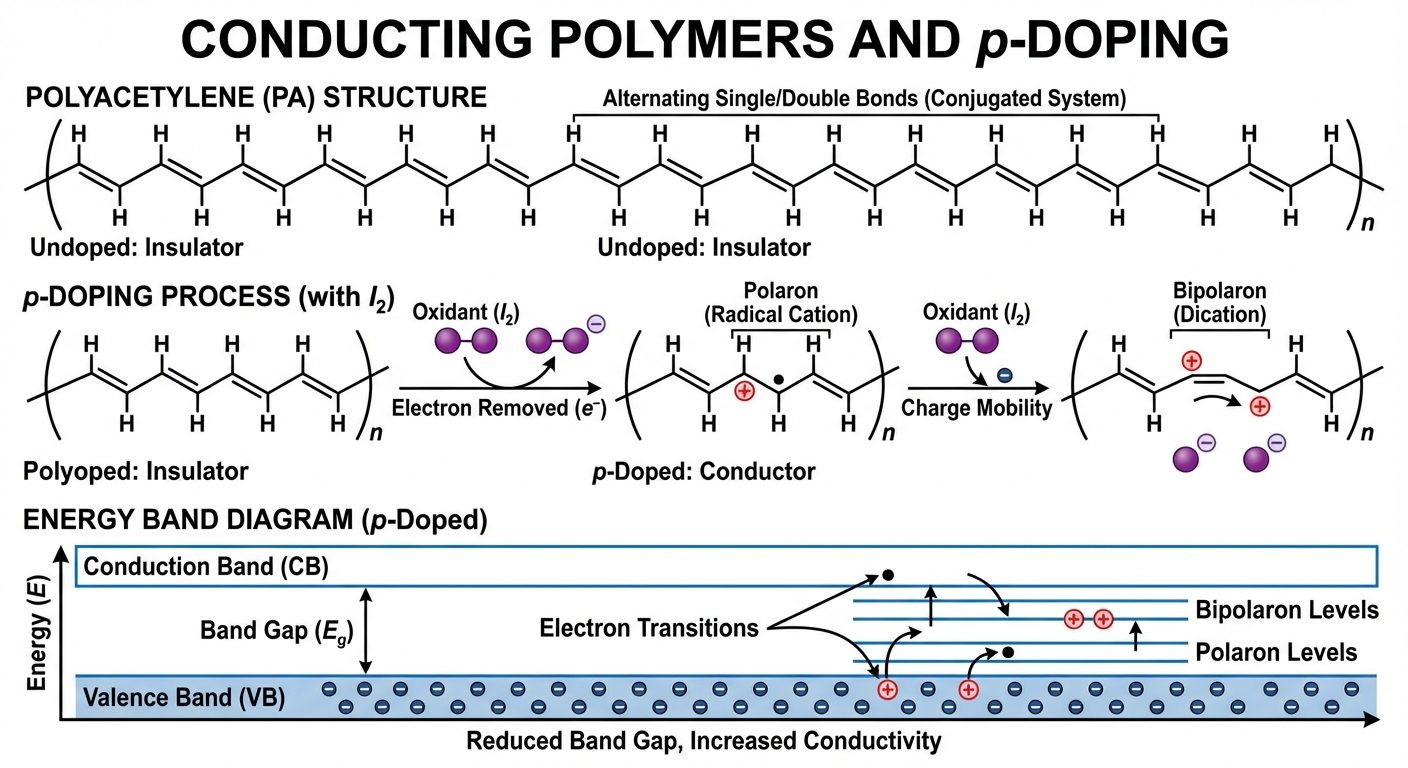
8. Conducting Polymers (ICPs)
Usually, organic polymers are insulators. Intrinsically Conducting Polymers (ICPs) possess electrical conductivity similar to metals/semiconductors.
Mechanism: Conjugation and delocalization
- Conducting polymers have a Conjugated Backbone: Alternating single () and double () bonds (e.g., ).
- -electron Delocalization: The overlap of p-orbitals allows electrons to move along the polymer chain, reducing the band gap between the Valence Band (VB) and Conduction Band (CB).
Doping Mechanisms
Conductivity is drastically increased (from to S/cm) by Doping (introducing impurities).
- p-doping (Oxidative): Removing electrons from the polymer backbone using oxidizing agents (). Creates positive charges (holes).
- n-doping (Reductive): Adding electrons to the polymer backbone using reducing agents (Na, Li). Creates negative charges.
Charge Carriers
When doped, structural deformations occur, creating specific charge carriers:
- Solitons: Associated with degenerate ground states (e.g., Polyacetylene). A domain wall separating two phases of orientation.
- Polarons: A radical ion (spin 1/2) associated with lattice distortion. Formed in non-degenerate ground states (e.g., Polypyrrole).
- Bipolarons: A di-cation or di-anion (spin 0) formed by the combination of two polarons. Responsible for high conductivity.
Conductivity and Transport
- Transport occurs via intra-chain movement (along the backbone via delocalization) and inter-chain hopping (jumping from one chain to another).
- Conductivity range: S/cm (Neutral) to S/cm (Doped).
Applications
- Electronics: OLEDs (Organic LEDs), flexible displays, transistors (OFETs).
- Energy: Polymer solar cells, electrode materials for supercapacitors and batteries.
- Sensors: Chemical sensors (conductivity changes upon exposure to specific gases/chemicals).
9. Lubricants
Lubrication and Purpose
Definition: A substance introduced between two moving surfaces to reduce friction.
Purpose:
- Reduce friction and wear.
- Dissipate heat (coolant).
- Prevent corrosion.
- Seal gaps (e.g., piston rings).
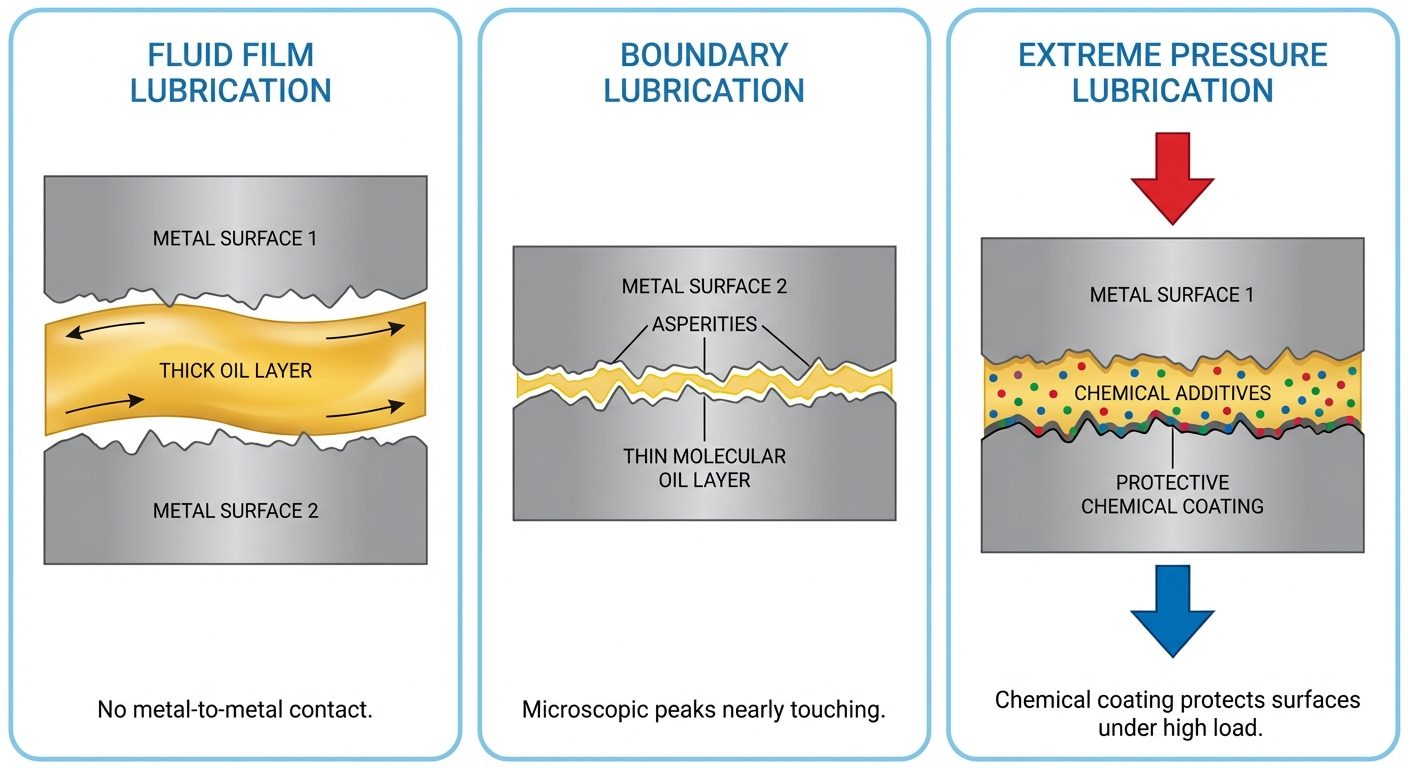
Mechanisms of Lubrication
- Fluid Film (Hydrodynamic) Lubrication: Thick oil film completely separates surfaces. Used in high speed, low pressure.
- Boundary Lubrication: Thin oil film (monolayer) adsorbed on surfaces. Asperities (surface peaks) may touch. Used in low speed, high pressure.
- Extreme Pressure (EP) Lubrication: Under high heat/pressure, additives react chemically with metal to form solid protective layers (chlorides/sulfides).
Additives for Lubricants
To improve performance, additives are mixed with base oils:
- Anti-oxidants: Prevent oxidation of oil (e.g., Aromatic amines).
- Anti-wear agents: Form protective films (e.g., ZDDP).
- Viscosity Index Improvers: Prevent oil from thinning at high temps (e.g., Polymethacrylates).
- Detergents: Keep engine parts clean from sludge.
Chemical and Physical Properties
- Viscosity: Resistance to flow. Most important property. Must be optimal (too low = friction; too high = energy loss).
- Viscosity Index (VI): Rate of change of viscosity with temperature. High VI is desirable (viscosity changes little with heat).
- Flash and Fire Point:
- Flash Point: Lowest temp where oil vapor ignites momentarily.
- Fire Point: Temp where oil burns continuously. (Safety indicators).
- Cloud and Pour Point:
- Cloud Point: Temp where oil becomes hazy (wax crystallization).
- Pour Point: Lowest temp where oil flows. (Important for cold weather operation).
- Aniline Point: Lowest temp at which oil mixes with aniline. High aniline point Low aromatic content (Good, prevents rubber seal damage).
- Neutralization Number (Acid/Base Value): Measures acidity/alkalinity. Indicates age/degradation of oil.