Unit 1 - Notes
Unit 1: Water and its Treatment
1. Hardness of Water
Hardness is the property of water that prevents the lathering of soap. It is caused by the presence of dissolved salts of heavy metals, primarily Calcium () and Magnesium (), and sometimes Iron (), Aluminum (), and Manganese ().
When hard water is treated with soap (sodium stearate), it forms a white precipitate (scum) instead of lather.
1.1 Types of Hardness
-
Temporary Hardness (Carbonate Hardness):
- Caused by the presence of dissolved bicarbonates of Calcium and Magnesium (, ).
- Removal: Can be removed simply by boiling.
- Reaction upon boiling:
-
Permanent Hardness (Non-Carbonate Hardness):
- Caused by chlorides and sulfates of Calcium and Magnesium ().
- Removal: Cannot be removed by boiling; requires chemical treatment (e.g., Lime-soda process, Zeolite process).
1.2 Units of Hardness
Hardness is usually expressed in terms of Calcium Carbonate () equivalents because its molecular weight is exactly 100 (easy calculation) and it is the most insoluble salt.
- Parts per million (ppm): Parts of equivalent hardness per parts of water.
- Milligrams per liter (mg/L): Mass of equivalent in milligrams per liter of water. ()
- Degree Clarke (): Parts of equivalent hardness per 70,000 parts of water.
- Degree French (): Parts of equivalent hardness per parts of water.
Relationship:
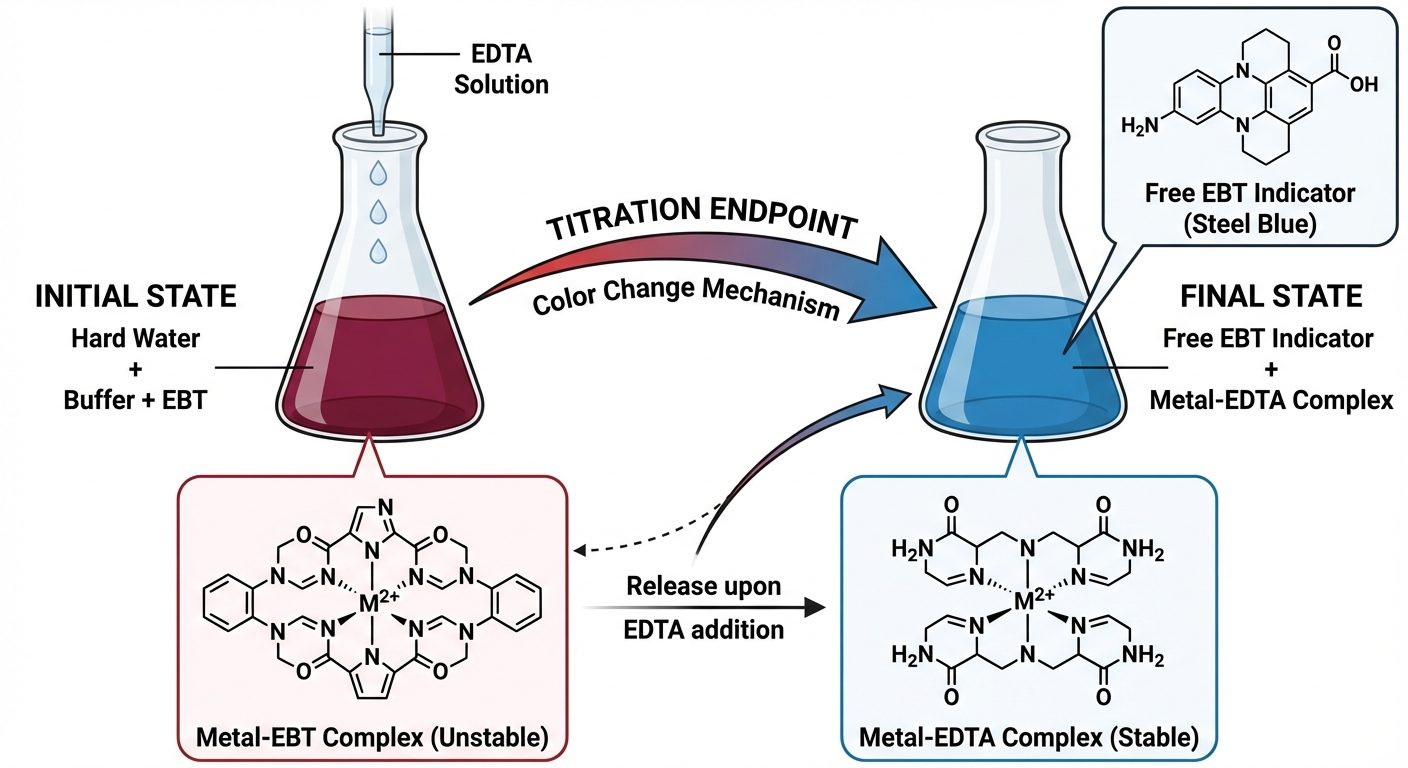
2. Determination of Hardness by EDTA Method
This is a complexometric titration method using Ethylene Diamine Tetra Acetic Acid (EDTA).
Principle
- Indicator: Eriochrome Black T (EBT).
- Buffer: (maintained at pH 10).
- Reagent: Disodium salt of EDTA.
When EBT is added to hard water at pH 10, it forms an unstable wine-red complex with ions. Upon titration with EDTA, the EDTA extracts the metal ions from the indicator complex (forming a stable colorless Metal-EDTA complex), releasing the free EBT indicator which is blue.
Reactions:
(End point: Wine Red to Steel Blue)
Calculation Formula
3. Alkalinity of Water
Alkalinity is the measure of water's ability to neutralize acids. It is primarily due to the presence of:
- Hydroxide ions ()
- Carbonate ions ()
- Bicarbonate ions ()
Determination
It is determined by titrating water against a standard acid using two indicators: Phenolphthalein (P) and Methyl Orange (M).
- P-End Point (pH > 8.3): Neutralizes all and half of .
- M-End Point (pH < 4.5): Neutralizes remaining half of and all .
Significance:
- High alkalinity causes caustic embrittlement in boilers.
- It influences the dosage of chemicals in coagulation and softening.
4. Boiler Feed Water and Troubles
Water used in steam generation boilers must be free from hardness and dissolved gases. Using raw water causes the following troubles:
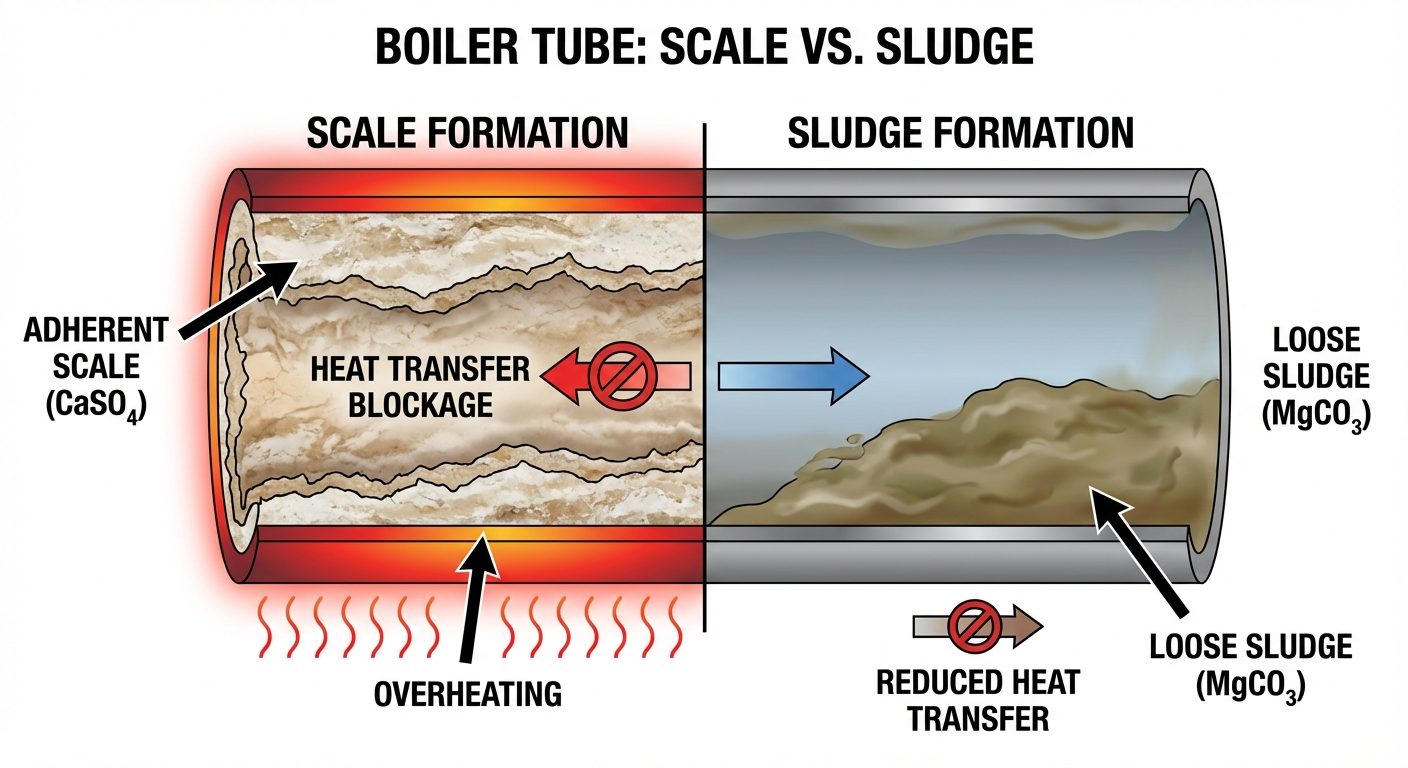
4.1 Sludge and Scale Formation
As water evaporates, the concentration of dissolved salts increases, leading to precipitation.
- Sludge: A soft, loose, and slimy precipitate (e.g., ). It settles in colder parts of the boiler and can be removed by "blow-down operation."
- Scale: A hard, adherent coating on the inner walls (e.g., ). It is a poor conductor of heat, causing fuel wastage and potential boiler explosion due to overheating.
4.2 Priming and Foaming (Carry Over)
- Priming: Rapid boiling causing wet steam (water droplets mixed with steam). Caused by high water levels or sudden steam demand.
- Foaming: Formation of persistent foam bubbles on the water surface. Caused by oils, alkalis, or dissolved organic matter.
4.3 Caustic Embrittlement
A type of boiler corrosion caused by high alkalinity. In high-pressure boilers, hydrolyzes to . The caustic soda concentrates in cracks/rivets, making the metal brittle due to the formation of sodium ferroate.
5. Softening Methods (Water Treatment)
5.1 Internal Treatment (Conditioning)
Chemicals are added directly into the boiler to prevent scale.
- Colloidal Conditioning: Kerosene or agar-agar is added to coat scale-forming precipitates, making them non-adherent (easy to blow-down).
- Phosphate Conditioning: is added to high-pressure boilers. It reacts with Calcium to form soft Calcium Phosphate sludge ().
- Calgon Conditioning: Sodium Hexametaphosphate (Calgon) forms soluble complexes with , preventing precipitation.
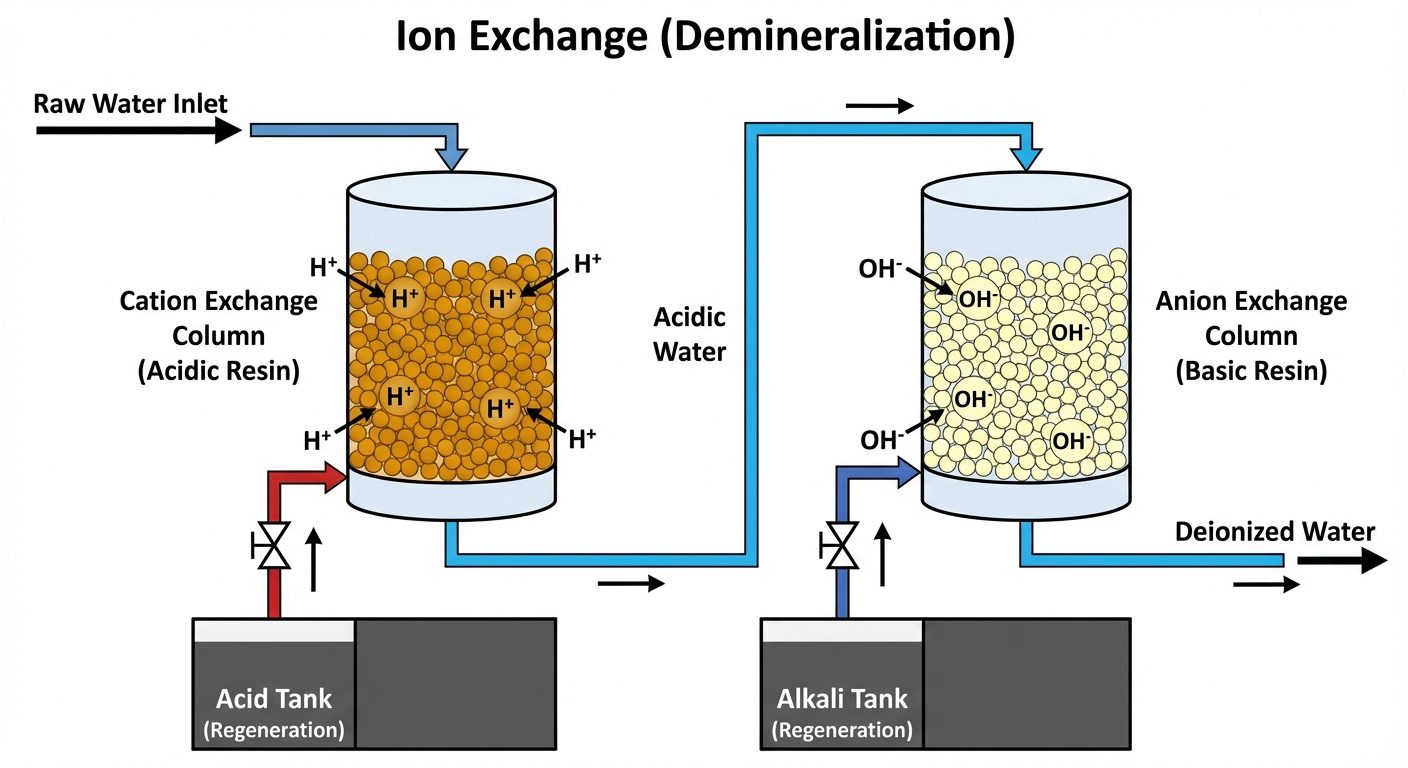
5.2 External Treatment: Ion Exchange (Demineralization) Process
This process removes almost all ions (cations and anions) to produce soft water (Demineralized Water).
- Cation Exchanger (): Resin containing acidic groups (). Exchanges for .
- Anion Exchanger (): Resin containing basic groups (). Exchanges for .
The from cation exchange and from anion exchange combine to form water ().
Regeneration:
- Cation resin: Dilute or .
- Anion resin: Dilute .
6. Numerical Problems: Concepts
To solve hardness numericals, convert all impurities into CaCO₃ equivalents.
Formula:
Or simply:
Common Multiplication Factors:
- (MW 162):
- (MW 146):
- (MW 136):
- (MW 120):
- (MW 111):
- (MW 95):
Steps for Calculation:
- Identify impurities.
- Calculate CaCO₃ equivalents for each.
- Temporary Hardness = Sum of CaCO₃ eq. of Bicarbonates.
- Permanent Hardness = Sum of CaCO₃ eq. of Chlorides & Sulfates.
- Total Hardness = Temporary + Permanent.
7. Membrane-Based Treatment Processes
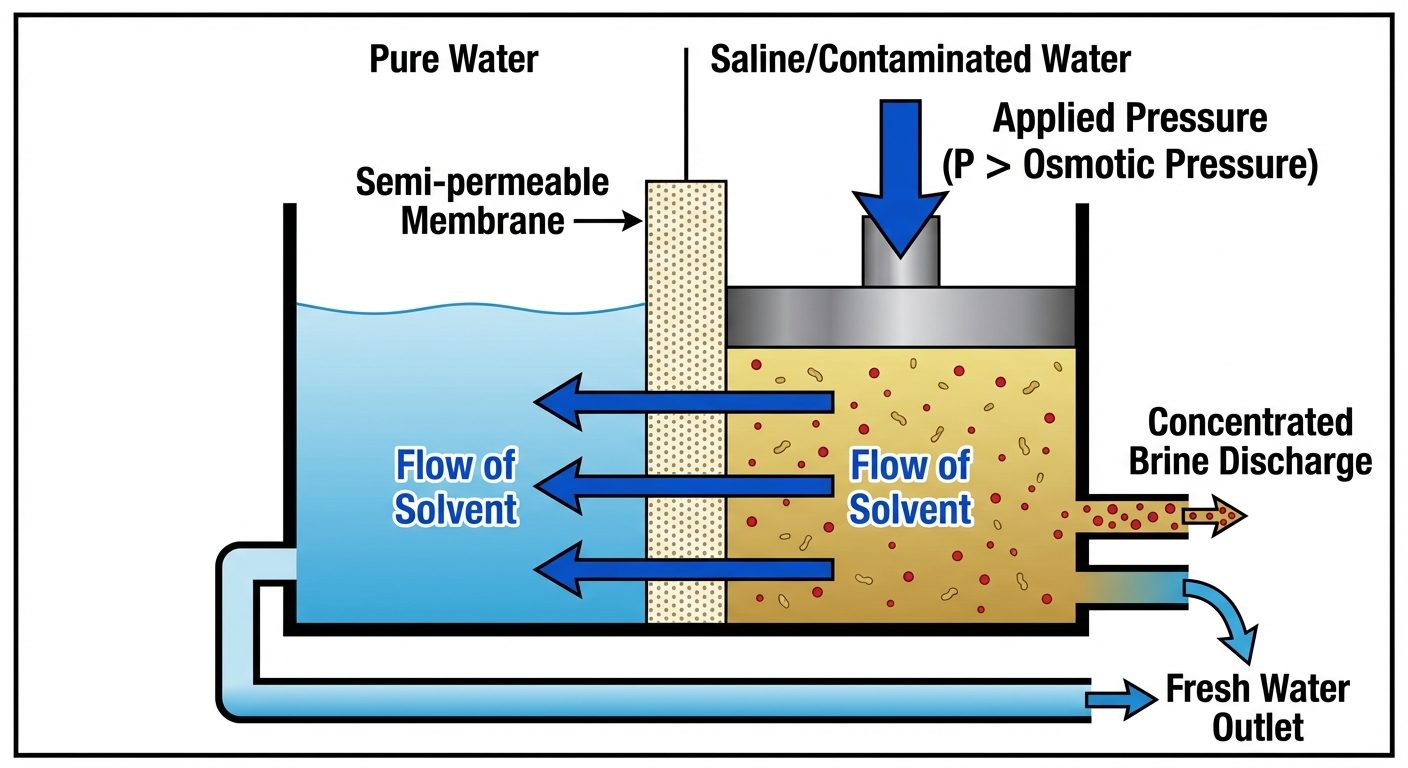
7.1 Reverse Osmosis (RO)
Usually, solvent flows from lower concentration to higher concentration through a semi-permeable membrane (Osmosis). If hydrostatic pressure greater than the osmotic pressure is applied on the concentrated side (impure water), the solvent flow reverses. Pure water is forced out of the salt solution.
- Membranes used: Cellulose acetate, Polyamide, Polysulfone.
- Advantages: Removes ionic salts, organic bacteria, and colloidal particles. Energy efficient.
7.2 Electrodialysis
Used for desalination of brackish water.
- Principle: Ions migrate towards electrodes of opposite charge under the influence of a Direct Current (DC) electric field.
- Setup: Consists of alternating Cation Selective Membranes (permeable only to cations) and Anion Selective Membranes.
- Result: Ions are removed from the central compartment, leaving fresh water.
8. Water Treatment by Chlorination
Disinfection is the process of killing pathogenic bacteria. Chlorination is the most common method.
Mechanism
Chlorine reacts with water to form Hypochlorous acid (), which is a powerful germicide.
enters the cell walls of bacteria and destroys the enzymes essential for their survival.
Break Point Chlorination
The point at which all reducing impurities and chloramines are oxidized, and free residual chlorine begins to appear.
- Stage 1: Chlorine kills microorganisms and oxidizes reducing impurities ().
- Stage 2: Chlorine forms Chloramines (with Ammonia).
- Stage 3: Destruction of Chloramines.
- Break Point: After this point, added chlorine remains as "Free Residual Chlorine," ensuring water safety against future contamination.
9. Specifications for Drinking Water (BIS 10500 : 2012)
Drinking water must be potable (safe to drink).
| Parameter | Desirable Limit | Permissible Limit (in absence of alternate source) |
|---|---|---|
| pH | 6.5 – 8.5 | No relaxation |
| Total Dissolved Solids (TDS) | 500 mg/L | 2000 mg/L |
| Total Hardness (as ) | 200 mg/L | 600 mg/L |
| Chlorides () | 250 mg/L | 1000 mg/L |
| Fluoride () | 1.0 mg/L | 1.5 mg/L |
| Nitrate () | 45 mg/L | No relaxation |
| Turbidity | 1 NTU | 5 NTU |
| E. coli / Coliform | Shall not be detectable in 100 ml sample | - |