Unit 4 - Notes
Unit 4: Quantum Mechanics
1. Need of Quantum Mechanics
By the end of the 19th century, Classical Mechanics (Newtonian mechanics) successfully explained the motion of macroscopic bodies (planets, projectiles, machinery). However, it failed to explain phenomena occurring at the atomic and sub-atomic levels.
Limitations of Classical Mechanics
Classical mechanics could not explain:
- Black Body Radiation: Classical theory (Rayleigh-Jeans Law) predicted infinite energy emission at high frequencies (the "Ultraviolet Catastrophe"), whereas experimental data showed intensity dropping to zero at very high frequencies.
- Photoelectric Effect: Classical wave theory suggested light intensity determines electron emission energy, but experiments proved frequency is the deciding factor.
- Atomic Stability: Maxwell's electromagnetic theory predicted that accelerating electrons should radiate energy and spiral into the nucleus, making atoms unstable.
- Line Spectra: Classical physics could not explain the discrete spectral lines of Hydrogen.
Conclusion: A new framework was required to treat energy and matter not as continuous flows, but as discrete packets (quanta). This gave birth to Quantum Mechanics.
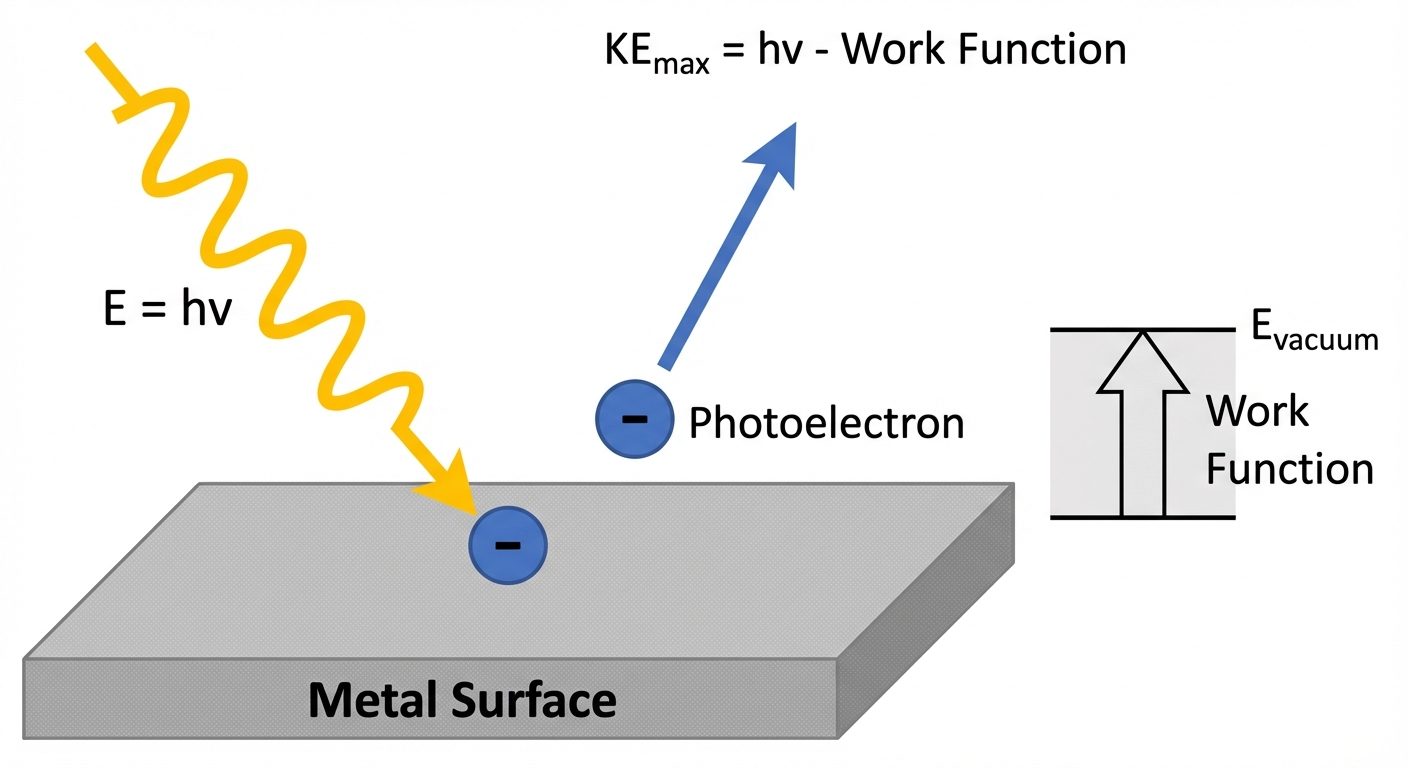
2. Photoelectric Effect
The phenomenon of emission of electrons from the surface of a metal when electromagnetic radiation (light) of suitable frequency strikes it.
Laws of Photoelectric Emission
- Threshold Frequency (): For a given metal, there exists a minimum frequency below which no emission occurs, regardless of intensity.
- Instantaneous Process: There is no time lag () between the incidence of light and emission of electrons.
- Kinetic Energy: The maximum kinetic energy of emitted photoelectrons is directly proportional to the frequency of incident light and is independent of intensity.
- Photoelectric Current: The number of photoelectrons emitted per second is directly proportional to the intensity of incident light.
Einstein’s Photoelectric Equation
Einstein applied Planck’s Quantum Theory, stating light consists of photons with energy . When a photon hits an electron:
Where:
- = Energy of incident photon.
- (Work Function) = Minimum energy required to pull the electron out of the metal surface ().
- = Maximum kinetic energy of the emitted electron.
Rearranging for kinetic energy:
3. Concept of de Broglie Matter Waves
In 1924, Louis de Broglie hypothesized that if radiation (light) can behave like a particle (photon), then material particles (like electrons, protons) should also behave like waves.
- Wave-Particle Duality: Matter has a dual nature. It manifests as a particle in some experiments (collisions) and as a wave in others (diffraction).
- Matter Waves: The waves associated with moving material particles.
de Broglie Wavelength Formula
For a photon:
Equating them: .
Replacing with particle velocity and mass :
Where:
- = de Broglie wavelength
- = Planck’s constant ()
- = Momentum of the particle
4. Wavelength of Matter Waves in Different Forms
The de Broglie wavelength can be expressed in terms of Energy, Voltage, and Temperature.
A. In terms of Kinetic Energy ()
Kinetic energy
Substituting in :
B. In terms of Accelerating Potential () for Charged Particles
If a particle with charge is accelerated through a potential difference , the work done equals kinetic energy:
Special Case: For an Electron
- kg
- C
- J·s
Substituting these values:
C. In terms of Temperature (Thermal Neutrons)
For gas molecules or neutrons at absolute temperature , the average kinetic energy is given by kinetic theory:
Where is the Boltzmann constant ().
5. Heisenberg Uncertainty Principle
Werner Heisenberg (1927) stated that it is physically impossible to measure both the position and momentum of a microscopic particle simultaneously with infinite accuracy.
Mathematical Statement
Where:
- = Uncertainty in position
- = Uncertainty in momentum ()
- (Reduced Planck's constant)
Energy-Time Uncertainty
Similarly, for energy and time:
Implication:
- If position is known precisely (), momentum becomes infinitely uncertain ().
- This principle explains why electrons cannot exist inside the nucleus (confinement in such a small space implies velocity greater than light, which is impossible).
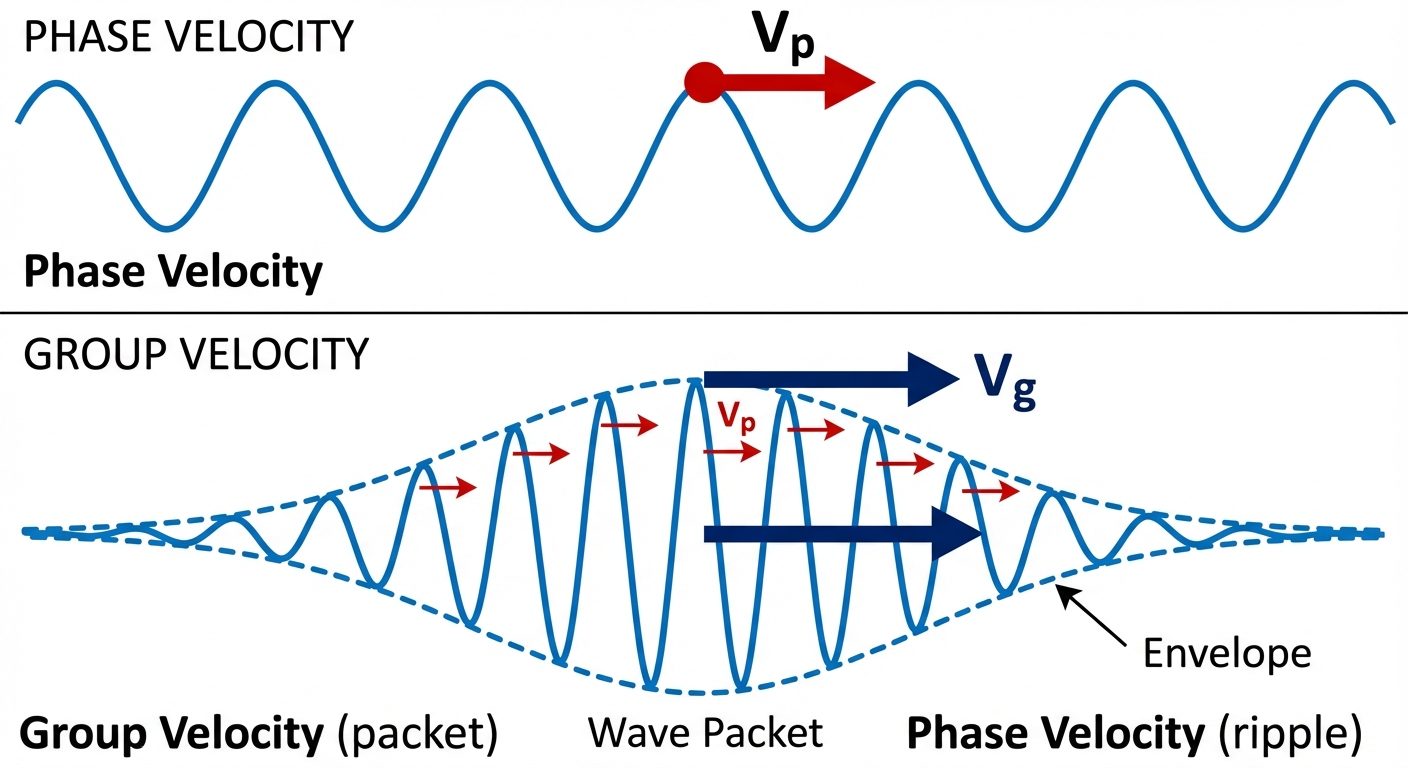
6. Phase Velocity and Group Velocity
Since particles behave like waves, we must define their velocity in wave terms.
Phase Velocity ()
- Also called Wave Velocity.
- It is the velocity with which a specific phase (like a single crest) of the wave travels.
- Formula:
- (Angular frequency)
- (Wave propagation constant)
- Issue: For matter waves, often calculates to be , which is greater than the speed of light (). This suggests a single wave cannot represent a particle.
Group Velocity ()
- A particle is represented by a Wave Packet (a group of waves with slightly different frequencies interfering constructively).
- Group velocity is the velocity with which the "envelope" or the packet travels.
- Formula:
- Relation to Particle Velocity: It is found that . The particle travels at the speed of the wave group.
Relation between and
- In a non-dispersive medium (like vacuum for light), .
- In a dispersive medium (matter waves), .
7. Wave Function and its Significance
In quantum mechanics, the state of a particle is described by a variable quantity called the Wave Function, denoted by .
Physical Significance
- No Direct Physical Meaning: itself is often a complex quantity () and cannot be measured directly.
- Probability Density: The quantity (or ) represents the probability of finding the particle at a specific point in space at a specific time.
- If is large: High probability of finding the particle.
- If : The particle is not there.
Characteristics of a Well-Behaved Wave Function
To be physically acceptable, must satisfy:
- Finite: must not be infinite anywhere.
- Continuous: and its first derivative must be continuous.
- Single-valued: At any point, there can be only one value of (only one probability possible).
- Normalized: The particle must exist somewhere in space.
8. Schrödinger Wave Equations
Erwin Schrödinger developed the fundamental differential equations that describe how the wave function changes over space and time.
A. Schrödinger Time-Dependent Wave Equation (STDE)
Describes a particle moving in a potential where total energy is conserved ().
Or, using the Hamiltonian operator ():
Where
B. Schrödinger Time-Independent Wave Equation (STIE)
Used for stationary states where Potential Energy () does not depend on time (). We separate the time and space parts.
For 1-Dimension ():
Where:
- = Space-dependent part of the wave function
- = Mass of particle
- = Total Energy
- = Potential Energy
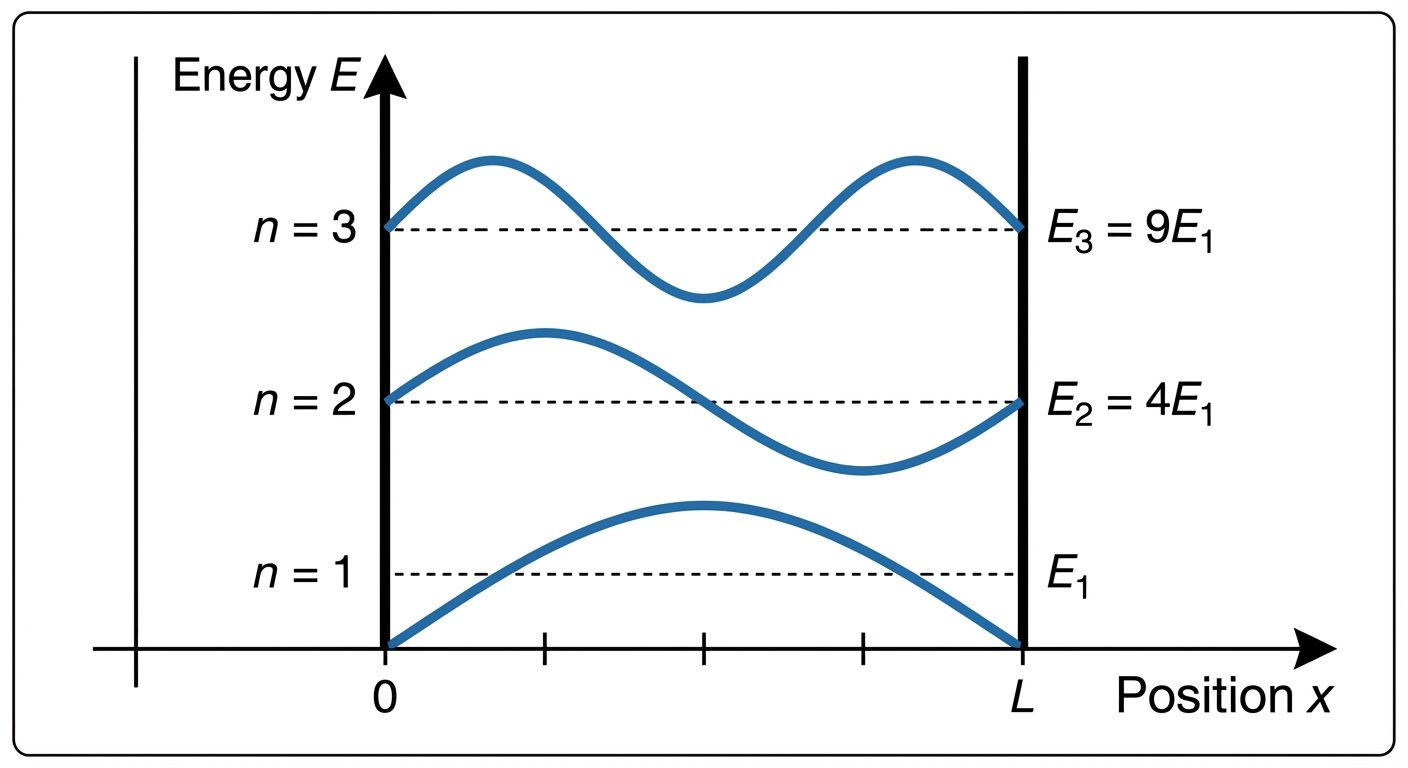
9. Particle in a One-Dimensional Box (Infinite Potential Well)
This is the simplest application of Schrödinger’s equation. Imagine a particle moving freely inside a box of length but cannot escape.
The Setup
- Region I (): (Particle cannot exist here).
- Region II (): (Free motion).
- Region III (): (Particle cannot exist here).
Solving the Equation
Inside the box (), the STIE becomes:
Let .
General solution:
Boundary Conditions
- At , .
- At , .
Since , .
Results
-
Eigenfunctions (Wave Functions):
After normalization ():
-
Eigenvalues (Energy Levels):
Substituting back into :
Conclusions
- Quantization: Energy is not continuous. It can only take discrete values ().
- Zero Point Energy: For , . The particle is never at complete rest.
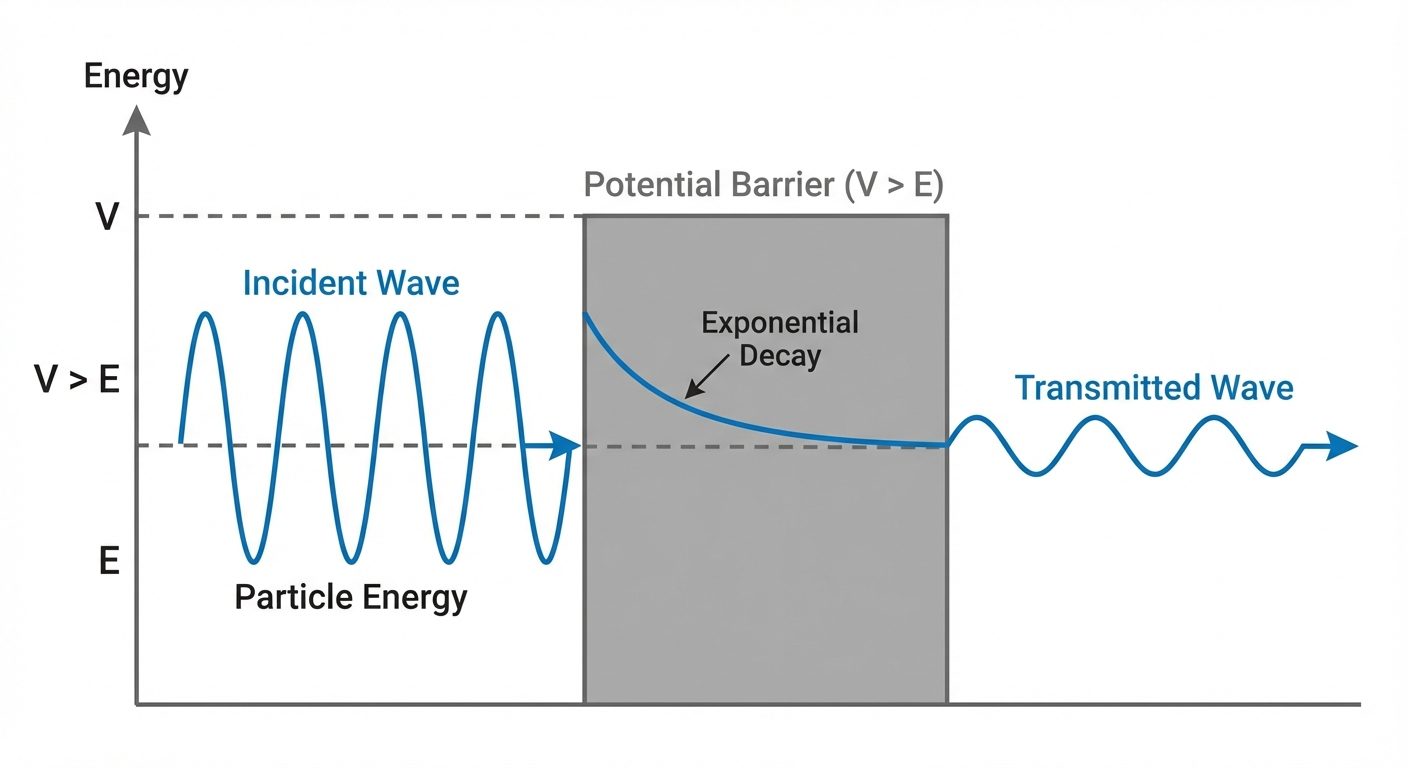
10. Tunneling Effect (Qualitative Idea)
In Classical Mechanics, if a particle approaches a potential barrier with height and the particle's energy , it will bounce back. It cannot cross the barrier.
In Quantum Mechanics, the wave function does not drop abruptly to zero at the barrier wall. Instead, it decays exponentially inside the barrier.
The Phenomenon
If the barrier is thin enough (width is small), the wave function may still have a non-zero value on the other side of the barrier.
- This implies there is a finite probability that the particle "tunnels" through the barrier, even though it lacks the energy to overcome it classically.
- Transmission Coefficient (): The probability of transmission. .
Applications
- Alpha Decay: How alpha particles escape the nuclear potential well.
- Tunnel Diode: Used in high-speed electronics.
- Scanning Tunneling Microscope (STM): Uses tunneling current to image surfaces at atomic resolution.